Express the adiabatic compressibility, in terms of measurable properties. Ks || 1 S
Question:
Express the adiabatic compressibility,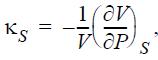
in terms of measurable properties.
Transcribed Image Text:
Ks || 1а ИӘР S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
To express the adiabatic compressibility Ks in terms of measurable properties we start with the defi...View the full answer

Answered By

Muhammad Salman Alvi
Well, I am a student of Electrical Engineeing from Information Technology University of Punjab. Just getting into my final year. I have always been good at doing Mathematics, Physics, hardware and technical subjects. Teaching profession requires a alot of responsibilities and challenges.
My teaching experience started as an home tutor a year ago. When I started teaching mathematics and physic subjects to an O Level student. He was about 14 years old. His name was Ibrahim and I used to teach him for about 2 hours daily. Teaching him required a lot of patience but I had to be polite with him. I used to give him a 5 min break after 1 hour session. He was quite weak in basic maths and calculation. He used to do quite a lot of mistakes in his homework which I gave him weekly. So I decided to teach him basics from scratch. He used to say that he got the concept even if he didn't. So I had to ask him again and again. I worked on his basics for a month and after that I started taking a weekly test sesions. After few months he started to improve gradually. Now after teaching him for about a year I can proudly say that he has improved alot. The most important thing was he managed to communicate all the difficullties he was facing. He was quite capable and patient. I had a sincere desire to help him reach to its full potential. So I managed to do that. We had a very good honest relationship of a student and a teacher. I loved teaching him as a tutor. Now having an experience of one year teaching I can read students quite well. I look forward to work as an online tutor who could help students in solving their all sort of difficulties, problems and queries.
4.90+
29+ Reviews
43+ Question Solved
Related Book For 

Introductory Chemical Engineering Thermodynamics
ISBN: 9780136068549
2nd Edition
Authors: J. Elliott, Carl Lira
Question Posted:
Students also viewed these Engineering questions
-
Express the Joule-Thomson coefficient in terms of measurable properties for the following a. Vander Waals equation b. An ideal gas.
-
Express the Joule-Thomson coefficient in terms of measurable properties for the following: (a) Van der Waals equation given in Example 6.6 (b) An ideal gas. Example 6.6 Accounting for T and Vimpacts...
-
Several attempts have been made to express the partial derivatives of the most common thermodynamic properties in a compact and systematic manner in terms of measurable properties. The work of P. W....
-
4. (a) (c) 6 -6 5 6 5 7 6 (b) (d) -5 6 6 -7 [6-5] 7-6
-
Give an example of a private solution to an externality. What is the Coase theorem? Why are private economic participants sometimes unable to solve the problems caused by an externality?
-
A company produces two products, A and B. The sales volume for A is at least 80% of the total sales of both A and B. However, the company cannot sell more than 100 units of A per day. Both products...
-
You just received an unexpected bonus at work of \($10,000.\) While you plan on using a bit of it to celebrate by purchasing all the accounting books you ever dreamed of, how much should you deposit...
-
The 2010 balance sheet of Marias Tennis Shop, Inc., showed $680,000 in the common stock account and $4.3 million in the additional paid-in surplus account. The 2011 balance sheet showed $715,000 and...
-
BRIGHTLIGHT INC. manufactures lamps and expects to sell 3 5 0 , 0 0 0 units next year at $ 2 1 per unit. Planned per - unit manufacturing costs at that level of production are as follows: Variable $...
-
(a) Prove (b) For an ideal gas along an adiabat, (P/P i ) = (T/ T i ) C P /R . Demonstrate that this equation is consistent with the expression from part (a). JP OT S || P TVOp
-
Express in terms of P and/or T . (OH) V T
-
Describe three analytical techniques for financial statement analysis.
-
What are the three principal fiduciary duties imposed on partners?
-
What is the dilemma for the landlord when a tenant abandons the premises during the term of the lease?
-
To whom are directors and officers duties owed?
-
Can a partner receive a salary from the firm? What is the real nature of a partners salary?
-
How would you suggest that responsibility for insuring the premises be allocated in a large shopping mall? Describe the most difficult aspects of the problem.
-
You expect to receive a payment of 1 million British pounds after six months. The pound is currently worth $1.60 (1 = $1.60), but the future price is $1.56 (1 = $1.56). You expect the price of the...
-
PC Contractors, Inc., was an excavating business in Kansas City, Missouri. Union Bank made loans to PC, subject to a perfected security interest in its equipment and other assets, including...
-
Write the charge and mass balances for dissolving CaF 2 in water if the reactions are CaF,(8) = Ca2+ + 2F Ca?+ + H,O = CAOH+ + H* Ca2+ + F = CaF+ CaF2(s) = CaF2(aq) F + H* = HF(aq) HF(aq) + F = HF,
-
Write charge and mass balances for aqueous Ca 3 (PO 4 ) 2 if the species are Ca 2+ , CaOH + , CaPO - 4 , PO 3- 4 , HPO 2 4 - , H 2 PO - 4 , and H 3 PO 4 .
-
Using activities, find the concentrations of the major species in 0.10 M NaClO 4 saturated with Mn(OH) 2 . Take the ionic strength to be 0.10 M and suppose that the ion size of MnOH + is the same as...
-
Does the Illinois law about interpreting insurance contracts, exclusionary clauses, and experimental treatments correct as a policy matter? Does it go too far in protecting insureds, get it just...
-
please provide and detail specific routes the carriers work on, do they provide mainline routes, regional services, feeder services etc. please research and discuss if you are not researching a...
-
Defence lawyers are always eager to prove lack of malice (i.e., that harm was not intended). Not only may it be necessary for a defence, but it can also affect the amount of damages if they lose the...

Study smarter with the SolutionInn App


