Recent research suggests the following equation of state, known as the PC-SAFT model. (a) Derive an expression
Question:
Recent research suggests the following equation of state, known as the PC-SAFT model.
(a) Derive an expression for Z.
(b) Derive the departure function for (U-Uig).
ηP = b; m = constant proportional to molecular weight; ai, bi are constants.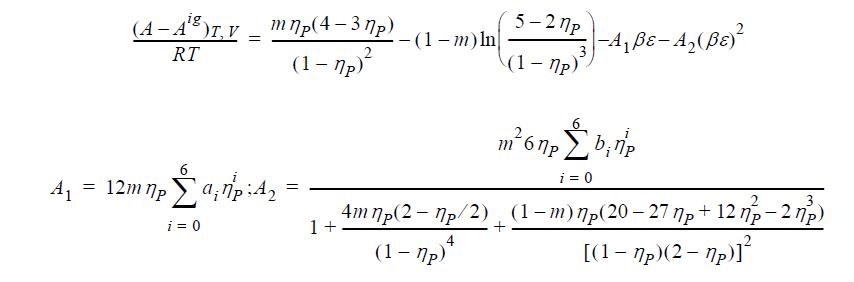
Transcribed Image Text:
(A - A°)T, V mnp(4-3np) ig. = RT 2 (1-7p)² = A₁ 12m np a₁7:4₂ i = 0 = -(1-m) ln 5-27p (1-7p) -A₁ßε-A₂ (ßε)² m²6 npb₁np i = 0 4 m np(2-7p/2) (1-m) mp(20-27 p + 12 np - 27p) 1+ (1-7p)* [(1-7p)(2-7p)]²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemical Engineering Thermodynamics
ISBN: 9780136068549
2nd Edition
Authors: J. Elliott, Carl Lira
Question Posted:
Students also viewed these Engineering questions
-
Derive an expression for the speed of sound based on van der Waals equation of state P = RT(v - b) - a/v2. Using this relation, determine the speed of sound in carbon dioxide at 50C and 200 kPa, and...
-
Derive an expression for the speed of sound based on van der Waals' equation of state P = RT(v - b) - a/v2. Using this relation, determine the speed of sound in carbon dioxide at 808C and 320 kPa,...
-
Recent research in thermodynamic perturbation theory suggests the following equation of state. (a) Derive the departure function for (A A ig ) T,V . (b) Derive the departure function for (U U ig )....
-
Using the 526 observations on workers in a certain dataset, we include educ (years of education), exper (years of labor market experience) and tenure (years with the current employer) in regression...
-
Draw the marginal-cost and average-total-cost curves for a typical firm. Explain why the curves have the shapes that they do and why they cross where they do.
-
Donald R. Cooper and C. William Emory discuss studying the relationship between on-the-job accidents and smoking. Cooper and Emory describe the study as follows: Suppose a manager implementing a...
-
Refer to Figure 8, the sequence diagram for the Make Appointment use case. Three spaces are identified in this diagram. Explain the significance of each.
-
James Banks was standing in line next to Robin Cole at Klecko's Copy Center, waiting to use one of the copy machines. "Gee, Robin, I hate this," he said. "We have to drive all the way over here from...
-
Q1 - At the heart of understanding marketing is something known as the marketing concept. In essence the marketing concept states: focus on creating transactions understand customer wants and needs...
-
P-V-T behavior of a simple fluid is found to obey the equation of state given in problem 8.14. (a) Derive a formula for the enthalpy departure for the fluid. (b) Determine the enthalpy departure at...
-
Suppose ethane was compressed adiabatically in a 70% efficient continuous compressor. The downstream pressure is specified to be 1500 psia at a temperature not to exceed 350F. What is the highest...
-
Given the information in Exercise 11.75, and using the 0.05 level of significance in comparing the sample standard deviations, were we justified in assuming that the population standard deviations...
-
If a citizen claims a provision in a statute to be unconstitutional because it is contrary to the Canadian Charter of Rights and Freedoms, must the government then show it is constitutional? Explain.
-
We have a parliament to pass laws, a government to administer laws, and a police department to enforce laws. Ironically, these potent instruments for the restriction of liberty are necessary for the...
-
What are the functions of regulatory bodies in safety assessment of nuclear reactors?
-
What are design extension conditions?
-
What is the probabilistic safety assessment (PSA) and its role in safety assessment of nuclear reactors. Explain the level of maturity achieved by this methodology over the years.
-
You have helped Marie establish financial goals and prepare a plan for achieving them. A tentative plan requires that Marie save $2,000 annually over the next five years. Since Marie has saved very...
-
The manager for retail customers, Katie White, wants to hear your opinion regarding one business offer she has received from an entrepreneur who is starting a mobile phone app called Easy Money. The...
-
What is the heat effect when 20 kg of LiCl(s) is added to 125 kg of an aqueous solution containing 10-wt-% LiCl in an isothermal process at 25C?
-
If a liquid solution of HCl in water, containing 1 mol of HCl and 4.5 mol of H 2 O, absorbs an additional 1 mol of HCl(g) at a constant temperature of 25C, what is the heat effect?
-
If LiCl2H 2 O(s) and H 2 O(l) are mixed isothermally at 25C to form a solution containing 10 mol of water for each mole of LiCl, what is the heat effect per mole of solution?
-
K Simplify by factoring. Assume that all variables under radicals represent nonnegative numbers. 36x6 Select the correct choice below and, if necessary, fill in the answer box that completes your...
-
Logan's Bakeshop makes cupcakes that cost $ 0 . 9 5 each. Logan knows that 2 0 % of the cupcakes will spoil. Assume Logan wants a 3 5 % markup on cost and produces 6 0 cupcakes. What should Logan...
-
Former Tongaat Hulett bosses in court for fraud Just over two years after charges were laid against them, former Tongaat Hulett Limited chief executive Peter Staude and six others accused of...

Study smarter with the SolutionInn App


