At a given temperature, pressure, and initial composition, the equilibrium mole fraction of species C for the
Question:
At a given temperature, pressure, and initial composition, the equilibrium mole fraction of species C for the following reaction:![]()
is found to be 0.75. What is the value of the mole fraction if the reaction is written as follows?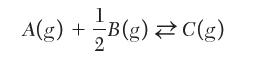
Transcribed Image Text:
2A(g) + B(g) 20(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
At low to moderate pressures the equilibrium state of the watergas shift reaction CO + H 2 O = CO 2 + H 2 is approximately described by the relation where T is the reactor temperature. K e is the...
-
A binary system of species l and 2 consists of vapor and liquid phase* in equilibrium at temperature T. The overall mole fraction of species I in the sy stem is d = 0.65. At temperature T. In 1 =...
-
The reaction N2 + O2 2NO occurs in internal combustion engines. Determine the equilibrium mole fraction of NO when the pressure is 101 kPa and the temperature is 1800 K.
-
There are 38 numbers in the game of roulette. They are 00, 0, 1, 2, . . ., 36. Each number has an equal chance of being selected. In the game, the winning number is found by a spin of the wheel. Say...
-
No major observatory has a Newtonian reflector as its primary instrument, whereas Newtonian reflectors are extremely popular among amateur astronomers. Explain why this is so.
-
To meet a sales quota, a car salesperson must sell 24 new cars, consisting of small, medium, and large cars. She must sell 3 more small cars than medium cars, and the same number of medium cars as...
-
You are applying for jobs at two companies. Company A offers starting salaries with = \($30,000\) and = \($4,000\). Company B offers starting salaries with = \($30,000\) and = \($2,000\). From...
-
Zander Consulting, a real estate consulting firm, specializes in advising companies on potential new plant sites. The firm uses a job cost system with a predetermined indirect cost allocation rate...
-
Brie recently finished her Business Program at Northwood Tech and was gifted $10,000 as a graduation gift. Her credit union offers special savings plans for students who finish Math with Business...
-
At 300 K and 1 bar, the equilibrium constant for the following reaction: is reported to be 10. What is the equilibrium constant at 300 K and 10 bar? You may assume ideal gas behavior. A(g) + B(g) C(g)
-
Consider the following reaction: The Gibbs energy of reaction at 298 K is determined to be and at a given temperature, the equilibrium constant is reported to be KT = 16. Now consider the reaction is...
-
How can a hotel sales representative identify who is responsible for purchasing meeting space, banquets, and rooms for corporate travelers in the corporate headquarters of an insurance company?
-
Justin Bieber has a tax rate of 35%. The par value for each of its bonds is $1,000. The bonds have 15 years to maturity and currently selling at 93% of their par value with a yield to maturity of...
-
What is a shared system of symbols, beliefs, and norms for behavior?
-
What is the purpose of administering magnesium sulfate to a patient in labor? What is the purpose of administering magnesium sulfate to a patient in labor? The purpose is to manage pain. The purpose...
-
How is the inflation we are experiencing today similar to the inflation the U.S. economy experienced after World War 2?
-
Coastal Company budgets sales of $1,280,000, fixed costs of $66,200, and variable costs of $294,400. What is the contribution margin ratio for Coastal Company? % If the contribution margin ratio for...
-
The program chair for the campus business club has asked you to present a talk to the group about consumer credit. He saw a newspaper article saying that only 10 percent of Americans know their...
-
Keating & Partners is a law firm specializing in labour relations and employee-related work. It employs 25 professionals (5 partners and 20 managers) who work directly with its clients. The average...
-
One of these isomeric alcohols has mp = 26C and bp = 82C; the other has mp = 90C and hp = 117C. Explain which isomer has the higher melting point and which has the higher boiling point.
-
Explain the differences in the boiling points between the members of each of these pairs of compounds: (a) CH 3 (CH 2 )6 CH3 bp: 126C CH3 (CH2)8 CH3 bp: 174C (b) CH3CH2CH2OH bp: 97C CH3CH2OCH3 bp:...
-
Explain the difference in the melting points of these isomers: mp=-140C 0 mp = 7C
-
How does the commodification of ethnic art impact the autonomy and agency of indigenous artists and communities, and what measures can be taken to address these concerns ?
-
5. Two approaches used to write the specifications for operations between a sender and a receiver object (e.g., Queue) are based on design by contract and defensive design. (a) Briefly describe each...
-
2. Find FIRST and FOLLOW for the following grammars: i) SabAB A bAB E BaBAa A E ii) S ABa A BD B be D de

Study smarter with the SolutionInn App


