Consider a gas that undergoes a process from state 1 to state 2. You know the ideal
Question:
Consider a gas that undergoes a process from state 1 to state 2. You know the ideal gas heat capacity and an equation of state. Which of the following hypothetical paths would be most appropriate to chose to calculate Δh? Explain.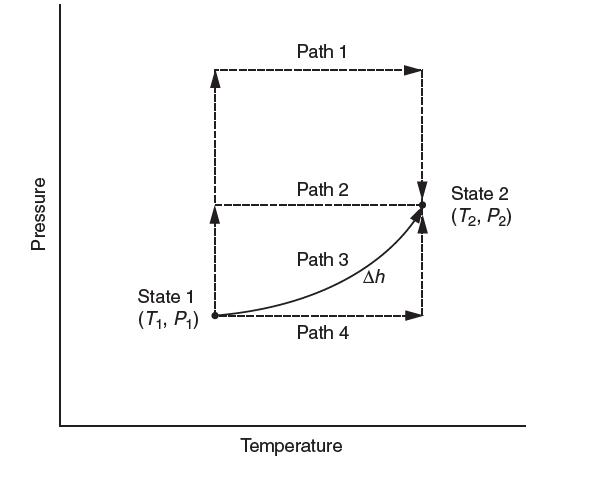
Transcribed Image Text:
Pressure State 1 (T, P) Path 1 Path 2 Path 3 Path 4 Temperature Ah State 2 (T2, P2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Rayan Gilbert
I have been teaching since I started my graduation 3 years ago. As a student, working as Teacher/PA has been tough but made me learn the needs for student and how to help them resolve their problems efficiently. I feel good to be able to help out students because I'm passionate about teaching. My motto for teaching is to convey the knowledge I have to students in a way that makes them understand it without breaking a sweat.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Consider a gas that undergoes a process from state 1 to state 2. You know the ideal gas heat capacity and an equation of state. Which of the following hypothetical paths would be most appropriate to...
-
An ideal gas undergoes a process from state 1 to state 2. For the following data, approximate the change in specific entropy for this process using Eq. 7.13. State T(C) P (kPa) 1 200 2 180 100 150...
-
An ideal gas undergoes a process from state 1 to state 2. For the data shown below, approximate the change in specific entropy for this process using Eq. 7.11. (ft lbm) 4.5 3.2 State T(F) P (psia) 1...
-
The United StatesMexicoCanada Agreement replaced what trade agreement?
-
LMX theory assumes that improved exchanges between leaders and followers are desirable. When might a follower not want "improved career-oriented social exchanges" with a leader? Describe one...
-
Burbank Corporation (calendar-year end) acquired the following property this year: Burbank acquired the copier in a nontaxable transaction when the shareholder contributed the copier to the business...
-
The same newbie changes the trial and runs the fan for a new reading. The calculations call for the trial weight to be increased from 100 to 400 g. What should the newbie do?
-
Visburg Concrete Company pours concrete slabs for single-family dwellings. Lancing Construction Company, which operates outside Visburg's normal sales territory, asks Visburg to pour 40 slabs for...
-
I had to go to the FBI office in Queens to do a hand analysis and sign my nana's (grandma) name numerous times and write out pages and pages of other sentences. I had no idea why because I never...
-
Consider the following property relation: (a) Come up with a physical process on a system which is described by the relation above. Sketch the process and describe it as completely as needed so that...
-
You are using the PengRobinson equation of state to determine the entropy change of an ideal gas: Is it better to try s = s (T,v) or s = s (T,P)? Explain. P = RT v-b aa(T) v(v + b) + b(v - b)
-
Three points are given in each of Problems 1114. Find the equation of the circle determined by these points, as well as its center and radius. (0, 0), (10,0), and (-7,7)
-
1. What is the principle of conservatism in accounting? How is it applied in financial reporting? 2. Explain the principle of materiality in accounting and how it influences financial...
-
The partners of Bentsen & Cole LLP had capital account balances of $45,000 and $25,000, respectively. They shared net income and losses in a 3:1 ratio. Prepare journal entries to record the admission...
-
For the fiscal year 2019-2020, Amazon was following straight line method of depreciation, but from the year 2021-2022, the company plans to use double declining method of depreciation. The Amazon's...
-
F= 3940 N with the projection angles as shown. F2=5910 N with the angles a -45, B = 60 and y =>90. (a) Knowing that y of F2 is greater than 90, what is the actual angle? (b) Express each of the two...
-
What are the three contributing factors that lead to people feeling sexually harassed in the workplace?
-
Describe three paragraph plans. Identify the uses for each.
-
Data on weekday exercise time for 20 females, consistent with summary quantities given in the paper An Ecological Momentary Assessment of the Physical Activity and Sedentary Behaviour Patterns of...
-
In this problem, we examine a model for the transport of oxygen from air in the lungs to blood. First, show that, for the initial and boundary conditions c(x, t) = c(x, 0) = co (0 < x < ] and c(0, t)...
-
Consult literature sources and list the observed timescales during which the following processes occur radiative decay of excited electronic states, molecular rotational motion, molecular vibrational...
-
Describe the main features, including advantages and disadvantages, of the following experimental methods for determining the rate law of a reaction: the isolation method, the method of initial...
-
A stock currently sells for $103.57 and is in the constant growth phase. The dividend yield is 5.93% and the capital gains yield is 2.80%. What will the stock's price be in 6 years? 2. A stock has a...
-
Q9. What is the price of a 6% bond in the following scenarios. The bond has 10 years The current rate of new bonds in the market is 4% Par Annual Coupon $ Rate Price (solve for Term present value) Is...
-
What will be the annual cash rate of a credit of S/10,000 granted for 1 month (30 days) to the TEA of 51.99. additionally applying a monthly credit life insurance cost of 0.0515% on the principal...

Study smarter with the SolutionInn App


