Consider a mixture of species 1, 2, and 3. The following equation of state is available for
Question:
Consider a mixture of species 1, 2, and 3. The following equation of state is available for the vapor phase: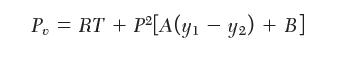
where,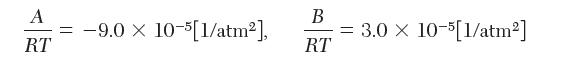
Transcribed Image Text:
P = RT + P [A(y - y) + B]
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Consider a mixture of species 1 and 2 in vaporliquid equilibrium at 25C and 90 bar. The following equation of state is available for the vapor phase: and y1 and y2 are the mole fractions of species 1...
-
Consider a mixture of species 1, 2, and 3. The following equation of state is available for the vapor phase: where, and y1, y2, and y3 are the mole fractions of species 1, 2 and 3, respectively....
-
A vaporliquid phase diagram for a binary mixture of species 1 and 2 at 293 K is shown in the following fi gure. Answer the following questions. (a) At 293 K, what is the value of P 1 sat ? (b)...
-
Migration is a popular strategy among many species. Monarch butterflies migrate between the Sierra Madre mountains in Mexico and many locations across the USA and Canada. Answer the following...
-
1- Briefly describe the background that led to the FBI initiation of the virtual case file (VCF) project. What do you think were the reasons why the VCF project failed? How do you relate your...
-
1. How is precedent created, and how is it applied in future cases? 2. When two separate panels of the same court hear different cases with similar issues, must the second panel follow the decision...
-
A linear viscoelastic, orthotropic lamina has principal creep compliances given in contracted notation by \[S_{i j}(t)=E_{i j}=F_{i j} t, \quad i, j=1,2, \ldots, 6\] where \(E_{i j}\) and \(F_{i j}\)...
-
A man buys a corporate bond from a bond brokerage house for $925. The bond has a face value of $1000 and pays 4% of its face value each year. If the bond will be paid off at the end of 10 years, what...
-
Category Land Land improvements Buildings Plant Asset $ 165,000 and Amortization $ 1,000,000 219,251 Equipment 625,000 307,500 Automobiles and trucks 162,000 90,325 Leasehold improvements 196,000...
-
A mixture of 2 moles propane (1), 3 moles butane (2), and 5 moles pentane (3) is contained at 30 bar and 200C. The van der Waals constants for these species are: Determine the fugacity and fugacity...
-
(a) Calculate the fugacity of pure methane vapor at T = 190.6 K and P = 32.2 bar. (b) Using the Lewis fugacity rule, calculate the fugacity of methane in a mixture of 80 mol% methane and 20 mol%...
-
Why did the FASB commence the Accounting Standards Codification_ project?
-
Given the production schedule above, what is the maximum number of workers the firm can hire before the effects of diminishing marginal returns set in?
-
describe the job of a manager for a business; include roles and responsibilities. Then, drawing on the concepts from operations management, discuss at least six (6) concepts that could apply as a...
-
describe several risk assessment methodologies you can draw upon to assess the risks and benefits associated with the management decision outcomes. In your description, address the following points:...
-
Describe several broad principles that could be applied in helping the individuals involved understand what went wrong in the following events. What suggestions could you make for remedying the...
-
Describe how you would adopt a balanced score card approach to evaluate your project performance. Your response should identify specific measures you would use as well as the approach you would take...
-
Discuss the brand development strategies marketers use to develop brands. Give an example of each strategy.
-
Hotel Majestic is interested in estimating fixed and variable costs so that the company can make more accurate projections of costs and profit. The hotel is in a resort area that is particularly busy...
-
Acetic acid was evaporated in container of volume 21.45 cm3 at 437 K and at an external pressure of 101.9 kPa, and the container was then sealed. The mass of acid present in the sealed container was...
-
The dissociation of I, can be monitored by measuring the total pressure, and three sets of results are as follows: T/K 973 1073 1173 100p/atm 6.244 7.500 9.181 104nj 2.4709 2.4555 2.4366 Where n1 is...
-
The 1980s saw reports of fH (SiH2) ranging from 243 to 289 k] mol-1. For example, the lower value was cited in the review article by R. Walsh (Ace. Chem. Res. 14,246 (1981)); Walsh later leant...
-
A competitor product supply issue would be ? Explain thoroughly.
-
Prepare a report form of balance sheet, assuming that the current portion of the note payable is $18,200. Enter account decreases and outflows as negative amounts.
-
Use symmetry to evaluate the following integral. 2 S (7-1)dx -2 2 S (7-1x1) dx=(Simplify your answer.) -2

Study smarter with the SolutionInn App


