Consider the isothermal mixing of 20% solute 1 by weight and 80% water, 2, at 25C. What
Question:
Consider the isothermal mixing of 20% solute 1 by weight and 80% water, 2, at 25°C. What is the heat transferred for the following mixtures?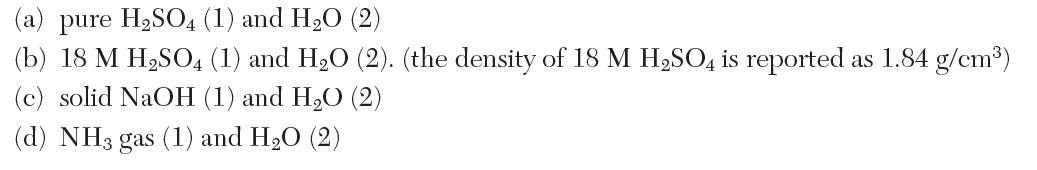
Transcribed Image Text:
(a) pure HSO4 (1) and HO (2) (b) 18 M HSO4 (1) and HO (2). (the density of 18 M HSO4 is reported as 1.84 g/cm) (c) solid NaOH (1) and HO (2) (d) NH3 gas (1) and HO (2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
Briefly describe ASCII and Unicode and draw attention to any relationship between them. [3 marks] (b) Briefly explain what a Reader is in the context of reading characters from data. [3 marks] A...
-
If an investor holds a credit-linked note and the credit event does not occur, the investor receives: A. All promised cash flows as scheduled. B. All coupon payments as scheduled but not the par...
-
Review the data model 1- Provide definitions of each of the following components: a- Entities b- Attributes c- Relationships d- Primary keys Identify each of the components from the data model above:...
-
Show that if \(x\) and \(z\) are positive real numbers and \(y\) is an arbitrary real number, the matrices form a group under matrix multiplication but the naive group integration measure \(d g=d x d...
-
What is the difference between the $t$ distribution and the normal distribution?
-
Fowler Company produces a product that sells for $200 per unit and has a variable cost of $125 per unit. Fowler incurs annual fixed costs of $450,000. Required a. Determine the sales volume in units...
-
Campbell, a single taxpayer, earns $400,000 in taxable income and $2,000 in interest from an investment in State of New York bonds. (Use the U.S. tax rate schedule.) Required: a. How much federal tax...
-
The following data are available for a binary mixture of ethanol and water at 20C: (a) Make a plot of the partial molar volumes of ethanol and water vs. mole fraction ethanol. (b) What is vmix for an...
-
Using data from Table 6.1, fi nd the partial molar enthalpy of water in a mixture of ethanol (1) and water (2) at 25C with x1 = 0.33. Use the same reference state as used in the steam tables. Steam...
-
Experience Hendrix, LLC, is owned by the sole heir of deceased rock guitar legend Jimi Hendrix. Experience Hendrix owns numerous trademarks associated with the musician, including the names "Hendrix"...
-
Are any of the following losses deductible on an individual's income tax return? If so, is the loss deductible "for" or "from" AGI? Explain each loss. Loss Loss on sale of stock in an individual's...
-
Rob Stevens is the chief executive officer of Isner Construction plc and owns 500,000 shares. The company currently has 4 million shares and convertible bonds with a face value of 20 million...
-
Suppose we are interested in testing the joint significance of b 1 and b 2 . That is, the null hypothesis is H 0 : 1 - 2 = 0. Test the joint significance of 1 and 1 .
-
Say that 90 percent of the people in a market demonstrate the endowment effect and 10 percent are rational. Say that, initially, all people have equal wealth. a. How would you expect the wealth...
-
Which of the following incorrectly shows the bond polarity? Show the correct bond polarity for those that are incorrect.
-
Pedigo Plastics Company accrued a tax liability for $4,000. Use the following horizontal financial statements model to show the effect of this accrual under the following two assumptions: (1) the tax...
-
San Carlos Bank and Trust Company uses a credit-scoring system to evaluate most consumer loans that amount to more than $2,500. The key factors used in its scoring system are found at the conclusion...
-
There is some indication that other hydrogen ring compounds and ions in addition to H3 and D3 species may play a role in interstellar chemistry. According to J.S. Wright and G.A. DiLabio (J. Phys....
-
Molecular orbital calculations may be used to predict trends in the standard potentials of conjugated molecules, such as the quinones and flavins that are involved in biological electron transfer...
-
Explain how a molecule is assigned to a point group.
-
Errors in Inventory Count Bow Corp. accidentally overstated its previous year ending inventory by $950. Assume that ending current year inventory is accurately counted. The error in the previous year...
-
For the year ended, December 31, 2020, the following information is available for the three product lines that Kincaid, Inc. carries in its inventory: Product Cost Market R89 $108,500 $106,900 N83...
-
Identify the instances of sexual harassment under Title VII of the Civil Rights Act of 1964. Multiple select question. Consensual relationships at the workplace Unwelcome sexual advances...

Study smarter with the SolutionInn App


