The following data are available for a binary mixture of ethanol and water at 20C: (a) Make
Question:
The following data are available for a binary mixture of ethanol and water at 20°C: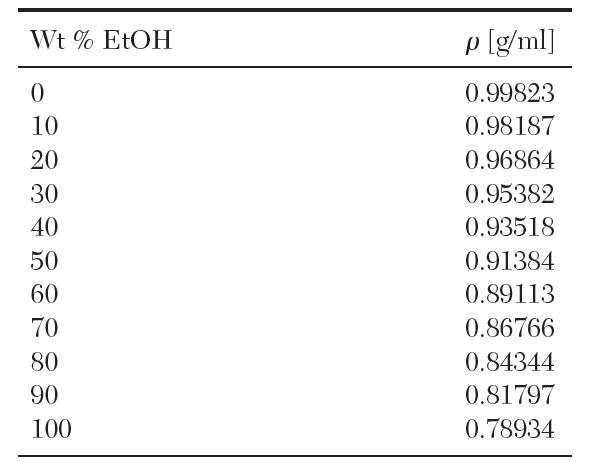
(a) Make a plot of the partial molar volumes of ethanol and water vs. mole fraction ethanol.
(b) What is Δvmix for an equimolar solution?
Transcribed Image Text:
Wt % EtOH 0 10 20 30 40 50 60 70 80 90 100 p[g/ml] 0.99823 0.98187 0.96864 0.95382 0.93518 0.91384 0.89113 0.86766 0.84344 0.81797 0.78934
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Older generations are more likely to view implanted microchips as a matter of convenience. True False
-
On April 15, Compton Co. paid $2,800 to upgrade a delivery truck and $125 for an oil change. Required: Journalize the entries for the upgrade to delivery truck and oil change expenditures.
-
Lanco Corporation, an accrual-method corporation, reported taxable income of $1,460,000 this year. Included in the computation of taxable income were the following items: MACRS depredation of...
-
Discuss the two-pipe system, how it works, and its advantages and disadvantages.
-
Provide a brief description about what type of data model you're developing, along with a project description.
-
If you were Buonannos manager, how would you have handled this situation? Think about the issue of an employee deciding not to accept a co-worker because of religious reasons. If you were the...
-
Show that for quasimonochromatic, stationary thermal light, the fourth-order coherence function \[ \boldsymbol{\Gamma}_{1234}\left(t_{1}, t_{2}, t_{3}, t_{4} ight)=E\left[\mathbf{u}\left(P_{1}, t_{1}...
-
Kristine bought a Rocky Mountain Chocolate Factory franchise. Her franchise agreement required her to purchase a cash register that cost $3,000, with an annual maintenance fee of $773. The agreement...
-
Lets say a portion of money that you add, usually, in a package at Quellenhof to cover the fee. Example: a hotel room costs 200 and the flight 100. Total 300. However, the guest did not book it...
-
The following data have been reported for the density, r, vs. mole fraction ethanol of binary mixtures of ethanol (1) and formamide (2) at 25C and 1 bar. X1 0 0.1000 0.1892 0.2976 0.3907 0.5009...
-
Consider the isothermal mixing of 20% solute 1 by weight and 80% water, 2, at 25C. What is the heat transferred for the following mixtures? (a) pure HSO4 (1) and HO (2) (b) 18 M HSO4 (1) and HO (2)....
-
Balance the following equation using the half-reaction method. H2O2+ ClO2 ClO2 + O2
-
Using the information below, what loan amount can be offered assuming a 9% debt yield? Round to nearest dollar: Gross Potential Income $ 1,500,000 Submarket Vacancy 3% Property Vacancy 7% Total...
-
During April, Burke Company has budgeted cash collections from customers of $68,000 and cash disbursements of $86,500. The budgeted ending Cash balances for March and April are $10,000 and $12,000,...
-
Scott received only stock of Little Grove Corporation in exchange for $15,000 cash and business equipment valued at $55,000. Immediately following the exchange, Scott owned 81% of the total combined...
-
Kelly Studio just bought two bikes for $3800 pre-tax. Kelly paid $190 in taxes, $60 for a 1 year warranty, and $150 in installation fees. Before the bikes were used for cycling, Kelly decided to pain...
-
On January 1, 2019, Maroon Corporation entered into a four-year operating lease. The payments were as follows: $20,000 for 2019, $18,000 for 2020, $16,000 for 2021, and $14,000 for 2022. What's the...
-
Document for Analysis: Weak Persuasive Memo Flowing Upward Your Task. Analyze the following memo, which suffers from many writing faults. List its weaknesses. If your instructor directs, revise the...
-
What are the principal differences among asset liquidity management, liability management, and balanced liquidity management?
-
An sp2 hybrid orbital that lies in the xy-plane and makes an angle of 1200 to the x-axis has the form Use hydrogenic atomic orbitals to write the explicit form of the hybrid orbital. Show that it has...
-
Derive eqns 11.11 and 11.14 by working with the normalized LCAO-MOs for the H+2 molecule-ion (Section 11.3a). Proceed by evaluating the expectation value of the Hamiltonian for the ion. Make use of...
-
We saw in Section 11.5 that, to find the energies of the bonding and antibonding orbitals of a heteronuclear diatomic molecule, we need to solve the secular determinant where aA et aB and we have...
-
Do you agree that supervising adults is similar to teaching children? Why or why not?
-
An inquiry-based learning strategy is a way to encourage inquiry during the learning process of the student. One important way to promote inquiry learning in a classroom is to design activities,...
-
Sethuraman Inc has the following periods of high and low costs and units produced during the period. Sethuraman Costs and Units Produced Month of Activity Units Produced Total Cost August 40,000...

Study smarter with the SolutionInn App


