Develop a general expression for ssys for an ideal gas that goes from (v1, T1) to (v2,
Question:
Develop a general expression for Δssys for an ideal gas that goes from (v1, T1) to (v2, T2) based on the path below.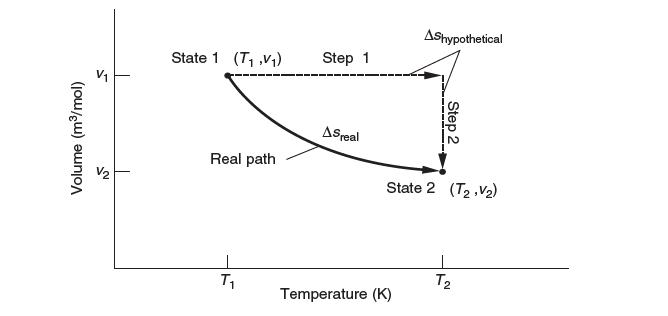
Transcribed Image Text:
S Volume (m/mol) NT State 1 (T.V) Real path T Step 1 ASreal AShypothetical Temperature (K) Step 2 State 2 (T, V) T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
An ideal gas flows adiabatically through a duct. At section 1, p1 = 140 kPa, T1 = 260°C, and V1 = 75 m/s. Farther downstream, p2 = 30 kPa and T2 = 207°C. Calculate V2 in m/s and s2 − s1...
-
A gas has an ideal gas heat capacity of C P * = (7/2)R and is described by the equation of state: Z = 1 + (CP 2 )/(RT) with C = 100 cm 3 /bar mol. A . Find a general expression for the residual...
-
The graph of f is given. (a) Why is f one-to-one? (b) What are the domain and range of f 1 ? (c) What is the value of f 1 (2)? (d) Estimate the value of f -1 (0). 1
-
Yates Company has a noncontributory, defined benefit pension plan. It is December 31, 1998, end of the accounting year and measurement date for the pension plan. The following are the data for three...
-
MGMT 6146 Module 6 Cconma.com in South Korea - An Innovative E-Commerce Platform provide summary of case ,problems issues recommendation and alternatives and also answer following question a. Among...
-
An unfinished concrete rectangular channel is \(5 \mathrm{~m}\) wide and has a slope of \(0.50^{\circ}\). The water is \(0.5 \mathrm{~m}\) deep. Find the discharge rate for uniform flow.
-
Bruce Albea Contracting, Inc., was the general contractor on a state highway project. Albea and the companies that agreed to guarantee the financial liabilities involved here (called sureties) agreed...
-
Job ordering costing system accumulates costs by individual product units. This method applies not only to individual inventory items but also batches of inventory items; It calculates manufacturing...
-
Develop a general expression for Dssys for an ideal gas that goes from (P1, T1) to (P2, T2) where heat capacity is given by: Cp = A + BT + CT2
-
The concept of entropy was developed in the nineteenth century, in order to study the efficiency of the steam engine, largely through the work of Sadi Carnot, Rudolph Clausius, and Lord Kelvin....
-
How are financial models superior to other screening models? How are they inferior?
-
How does speculation examination and portfolio the board affect monetary dynamic for a situation study, and what methods can be utilized to fabricate and improve a broadened venture portfolio for...
-
Refer to Table 10-4. a. What was the settlement price on the June 2020 5-Year U.S. Treasury Notes futures contract on March 13, 2020? (Do not round your intermediate calculations. Round your...
-
How does California tax capital gains? California applies a higher tax rate for capital gains. California applies a lower tax rate for capital gains. California does not tax capital gains. California...
-
Using coils, turbine, or compressor, to draw a simple flow diagram for a typical power production cycle and an air conditioning or refrigeration cycle, respectively. Then, redraw the air conditioning...
-
How does capital planning add to corporate money dynamic for a situation study, and what strategies are usually used to assess potential long haul speculation projects?
-
Olin Company currently makes only cash sales. Given the number of potential customers who have requested to buy on credit, Olin is considering allowing credit sales. What factors should Olin consider...
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
When D-galactose was heated at 165C, a small amount of compound A was isolated:
-
Phlorizin is obtained from the root bark of apple, pear, cherry, and plum trees. It has the molecular formula C21H24O10 and yields a compound A and D-glucose on hydrolysis in the presence of emulsin....
-
Emil Fischers determination of the structure of glucose was carried out as the nineteenth century ended and the twentieth began. The structure of no other sugar was known at that time, and none of...
-
Explain how do you plan to apply strategic process in your future professional endeavors?
-
What is the importance of writing minutes of the meeting? 2. What will happen if a company does not write minutes of the meeting every meeting? 3. What is the most difficult part in writing the...
-
Describe and explain what project documents should be archived?

Study smarter with the SolutionInn App


