Table 4.3 compares the van der Waals (1873), RedlichKwong (1949), and PengRobinson (1976) equations of state in
Question:
Table 4.3 compares the van der Waals (1873), Redlich–Kwong (1949), and Peng–Robinson (1976) equations of state in similar forms. Based on intermolecular interactions, qualitatively analyze how the progression of equations may have given more accurate results.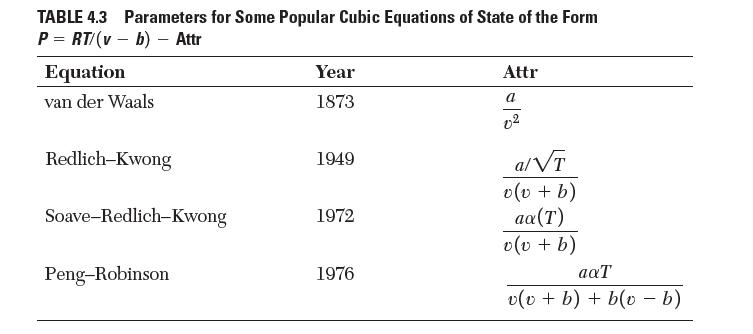
Transcribed Image Text:
TABLE 4.3 Parameters for Some Popular Cubic Equations of State of the Form P = RT/(vb) - Attr Equation van der Waals Redlich-Kwong Soave-Redlich-Kwong Peng-Robinson Year 1873 1949 1972 1976 Attr a a/VT v (v + b) ax (T) v (v + b) aaT v(v + b) + b(v - b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Van der Waals Equation and Critical Points (a) In p V- diagrams the slope p/V along an isotherm is never positive. Explain why. (b) Regions where p/V = 0 represent equilibrium between two phases;...
-
The van der Waals equation of state, an approximate representation of the behavior of gases at high pressure, is given by Where a and b are constants having different values for different gases. (In...
-
Table 10.3 shows that the van der Waals b parameter has units of L/mol. This means that we can calculate the sizes of atoms or molecules from the b parameter. Refer back to the discussion in Section...
-
Three charges are arranged as shown in the figure. Find the (a) magnitude and (b) direction (angle with the x axis) of the electrostatic force on the 6.00- nC charge. In the figure, q= 3.64 nC. 0.500...
-
What is the bookkeeping entry for a withdrawal of owner's capital?
-
Show that the fermion operators \(\hat{P}^{\mu}\) and \(\hat{M}^{\mu u}\) are the generators of the Poincar group by proving Eqs. (6.3.214), (6.3.212) and (6.3.213). Show that \(\hat{P}^{\mu}\) and...
-
The Durbin-Watson statistic is designed to detect autocorrelation and is defined by \[D W=\frac{\sum_{t=2}^{T}\left(y_{t}-y_{t-1} ight)^{2}}{\sum_{t=1}^{T}\left(y_{t}-\bar{y} ight)^{2}} .\] a. Derive...
-
United Furniture buys reclining rocking chairs at $550 less 40% and 10%. The price is marked up to allow for overhead of 50% of cost and profit of 35% of cost. The unit on display in the store...
-
Kendra, Cogley, and Mei share income and loss in a 3:2:1 ratio (in ratio form: Kendra, 3/6; Cogley, 2/6; and Mei, 1/6). The partners have decided to liquidate their partnership. On the day of...
-
At very high temperatures, a gas can be ionized and remain in thermodynamic equilibrium. Consider the case of gas containing only ions, A1. Your supervisor requests that you come up with a simple...
-
The second virial coeffi cient for argon is reported versus temperature in the following table. Explain the trend with temperature in terms of dominant intermolecular interactions. What can you say...
-
Build a simple Rails application that returns a static document to a requesting browser, where the static document is a brief description of you.
-
In what way are customers a source of information about a product, its competitive products, and the industry?
-
List the elements that a salesperson can use in planning and developing an informative presentation.
-
Can salespeople be motivated internally as well as by external rewards?
-
Describe how a salesperson can avoid information overload and explain why this is important.
-
Describe the concept of sales management and the role of the sales manager.
-
Manufacturing cost flow across three accounting cycles The following accounting events affected Gitau Manufacturing Company during its first three years of operation. Assume that all transactions are...
-
You purchase a bond with a coupon rate of 6.7 percent, a par value $1,000, and a clean price of $905. Assume a par value of $1,000. If the next semiannual coupon payment is due in two months, what is...
-
The p-toluenesulfonate shown undergoes an intramolecular Williamson reaction on treatment with base to give spirocyclic ether. Demonstrate your understanding of the terminology used in the preceding...
-
All the following questions pertain to 1H NMR spectra of isomeric ethers having the molecular formula C5H12O. (a) Which one has only singlets in its 1H NMR spectrum? (b) Along with other signals,...
-
The 1H NMR spectrum of compound A (C8H8O) consists of two singlets of equal area at 5.1 (sharp) and 7.2 ppm (broad). On treatment with excess hydrogen bromide, compound A is converted to a single...
-
Describing the level of engagement of the sales team in the company: There are 35 people that make up the sales team at XYZ Company and are generally happy with their employment. While they love...
-
Describe a recent organizational change in your organization (Google) that affected you. Extensively explain how would you discuss the leadership models , if there was no apparent leader in the...
-
Lisa is a supervisor working in Line CD-6 with 30 workers reporting to her. Her result-oriented and autocratic style has impressed her section ever since she joined the section. One Monday morning...

Study smarter with the SolutionInn App


