The following data are available for vaporliquid equilibrium of the methane (1) n-pentane (2) binary system at
Question:
The following data are available for vapor–liquid equilibrium of the methane (1) –n-pentane (2) binary system at 37.78°C. Compare how well the van der Waals and Peng–Robinson equations can represent these data using the equation of state method to calculate fugacity coeffi cients of the vapor and the liquid.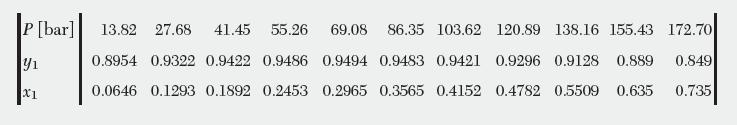
Transcribed Image Text:
P [bar] 13.82 27.68 41.45 55.26 69.08 86.35 103.62 120.89 138.16 155.43 172.70 0.849 0.8954 0.9322 0.9422 0.9486 0.9494 0.9483 0.9421 0.9296 0.9128 0.889 0.0646 0.1293 0.1892 0.2453 0.2965 0.3565 0.4152 0.4782 0.5509 0.635 0.735 y X1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
We can use the solution approach developed in Examples 815 and 816 to calculate the value of pressur...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Repeat Example 8.17 for the PengRobinson equation with values of the binary interaction parameter, k12 of 0.025, 0.05, and 0.10. Example 8.17 The following data are available for vaporliquid...
-
Compute the CLV metric and then apply it in a simple breakeven analysis. The data below show the results of a Starbucks segmentation study. Each column shows the values for a typical customer in each...
-
The directors of Mylo Ltd are currently considering two mutually exclusive investment projects. Both projects are concerned with the purchase of new plant. The following data are available for each...
-
Prepare adjusting journal entries, as needed, considering the account balances excerpted from the unadjusted trial balance and the adjustment data. A. depreciation on fixed assets, $ 8,500 B....
-
Which of the following questions should a marketing researcher ask when trying to establish the reliability of secondary data sources in the international arena? a. How much does the data cost? b....
-
List three examples of situations where this may be appropriate.
-
What is the maximum price an investor will pay for a bond with a face value of $\$ 5,000$ and a coupon rate of $12 \%$, paid annually? Assume the investor seeks a yield of $10 \%$ and the bond will...
-
Presented below are the purchases and cash payments journals for Rosalez Co. for its first month of operations. In addition, the following transactions have not been journalized for July. The cost of...
-
Given the following information, calculate the weighted average cost of capital for Puppet Corporation. (Round Intermediate calculations to 2 decimal places. Round the final answers to 2 decimal...
-
Calculate the equilibrium composition of the two liquid phases in a binary mixture of methyl diethylamine (a) and water (b) at 1 bar and 20C. Solve this problem in two ways; by graphical methods and...
-
Repeat Example 8.15 using the PengRobinson equation of state. Compare your answer to the reported measured value of P = 69.1 bar and y1 = 0.95. Example 8.15 Using the equation of state method,...
-
1. Consider the following savings account statement: (a) What interest rate is this bank paying? (b) Give the interest and balance on 4/1/17. (c) Give the interest and balance on 1/1/19. 2. Consider...
-
Let \(\left(B_{t}ight)_{t \geqslant 0}\) be a \(\mathrm{BM}^{1}\). Decide which of the following processes are Brownian motions: a) \(X_{t}:=2 B_{t / 4}\); b) \(Y_{t}:=B_{2 t}-B_{t}\); c)...
-
Best Trust has grown to one of the worlds top 25 banks by building on its broad name recognition and reputation for integrity. Its 73,000-employee workforce spans 47 countries. One of those employees...
-
The barriers to entry are so high in the automotive industry that it is rare to see a new entrant. A notable exception has been Malaysias PRGA Motors, the countrys largest maker of commercial...
-
What countries are negotiating to join the Trans Pacific Partnership (TPP)? What countries may be interested in joining this group? (Use information from the U.S. Trade Representative at www.ustr.gov...
-
A study of the effects of youth soccer programs on self-esteem found that children who played competitive soccer had more self-esteem than other children. Identify the most important statistical bias...
-
Not-For-Profit Financial Statements. Renfrow Rehabilitation Center uses fund accounting for internal purposes. Presented is the December 31, 2011 balance sheet prepared from the funds the center...
-
Velshi Printers has contracts to complete weekly supplements required by fortysix customers. For the year 2018, manufacturing overhead cost estimates total $600,000 for an annual production capacity...
-
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it. : a) H_C_C,H 4 (both) : ...
-
Circle the conjugated pi bonds, if any, in the following compounds? c) CH,CH=CH-C=N b) CH3-CH=CH-C-CH3 d) H-C=C-CH=CH-C-H La e) CH-CH=CH-CH-C-CH3
-
What is the hybridization at the indicated atoms in these compounds? a) CHCH=CH_NHCH3 12 3 c) CH,=CH0CH, 1 2 3 4 5 0: b) CHC0CH, 1 d) 2 1 NH
-
A pre paid cellular phone 25$ for activation and 0.05$ per minute. The relation of cost to minutes can be defined by the function c(x)=o.o5x + 25. What is the inverse function.
-
A steamroller has a large, heavy cylindrical barrel that is 4 m wide and has a diameter of 2 m. a Find the area of the curved surface of the barrel, to two decimal places. b After 10 complete turns...
-
5. Lucia and Suraya decide to purchase a house together as an investment property. They a. purchased the home for $752,000. After selling their Bit Coin stocks and pooling their savings they were...

Study smarter with the SolutionInn App


