Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of
Question:
Show the hybridization at each of the atoms, except H, in these molecules. Indicate the type of each designated bond and the orbital's that are overlapping to form it.
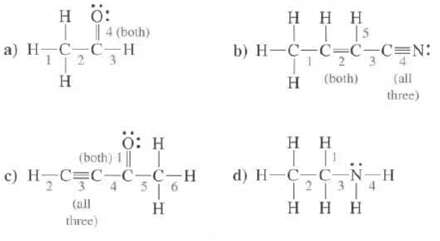
Transcribed Image Text:
Η Ο: a) H_C_C,H Η 4 (both) Ο: Η (beth)1] | 0 H_C_C_C=CH 4 16 Η Caill three) Η Η Η LIT b) H-C_C_C_C-N: H (both) Η Η Τ d) H=C=C, 1²1³1 Η Η Η 213 :NIH (all three) H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a 1 OCsp3 H1s b 1 OCsp3 Csp2 4 OCsp Nsp 2 OCsp3 Csp2 3 OCsp2 ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In the hydrocarbon (a) What is the hybridization at each carbon atom in the molecule? (b) How many Ï bonds are there in the molecule? (c) How many Ï bonds? (d) Identify all the 120o bond...
-
Indicate the type of atomic orbitals that are overlapping to form each of the different kinds of bonds in CH3OCH3 (For example, a carbon sp3 AO and a hydrogen Is AO). What kinds of orbitals are...
-
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds? ITT H=C=C=C=C_C7H (both) H tall...
-
The toroid of FIGURE P29.55 is a coil of wire wrapped around a doughnut-shaped ring (a torus). Toroidal magnetic fields are used to confine fusion plasmas. a. From symmetry, what must be the shape of...
-
lnkco manufactures printing inks. The company employs 150 people, of whom 120 are productt0 The ink market is extremely competitive and, with a 20 per cent market share, lnkco has the third largest...
-
In 2011, home prices and mortgage rates fell so far that in a number of cities the monthly cost of owning a home was less expensive than renting. The following data show the average asking rent for...
-
Here's a tough one. For the same data where two groups are being compared, the critical value for rejection of the null hypothesis is higher for a set of dependent groups than for a set of...
-
Irwin Corporation has been authorized to issue 20,000 shares of $100 par value, 10%, noncumulative preferred stock and 1,000,000 shares of no-par common stock . The corporation assigned a $2.50...
-
You have a Microsoft 365 E5 subscription that uses Microsoft Purview Communication Compliance. You need to create a notice template that supports the use of rich formatting. What format should you...
-
Visit two web sites that you are familiar with and write down every click that you make, and the patterns and time periods corresponding to your surfing. Then, describe how a marketerof a product of...
-
Consider hydrogen cyanide, H C N. (a) What is the hybridization at the N at the C? (b) What are the types of the three CN bonds? What orbitals overlapping to form them? (c) In what type of orbital...
-
Circle the conjugated pi bonds, if any, in the following compounds? c) CH,CH=CH-C=N b) CH3-CH=CH-C-CH3 d) H-C=C-CH=CH-C-H La e) CH-CH=CH-CH-C-CH3
-
Skyler Company sells a product for $120 per unit. The variable cost is $84 per unit, and fixed costs are $1,890,000. Determine (a) The break-even point in sales units and (b) The sales units required...
-
In 2023, Desi's Desserts had four employees. Two of the employees worked full time and earned salaries of $35,000 each. Two employees worked only part time, and each earned $5,000. The employer...
-
Staying safe during deployment requires preparation and planning. The following preparatory steps can help you stay safe during deployment. (Select the missing action.) Find out the local conditions...
-
Discussion Question: Envisioning the Future & Enlisting Others - CLO 2, CLO 3, CLO 4, CLO 5 What can leaders do to help others in the organization "see" the future? How can leaders help organizations...
-
63. The above figure shows the marginal private cost curve, marginal social cost curve, and marginal social benefit curve for raising goats on a common pasture. A quota to prevent the overuse of the...
-
Using the relationship a = exp (ln (a)) -- for example, in Audit Class 1, a = e 6.13 = 459 -- we can determine the equation for Normalized Cumulative ATR for each audit class as a function of the...
-
If forces always come in interaction pairs and the forces in such a pair are equal in magnitude and opposite in direction (Eq. 8.15), how can the vector sum of the forces exerted on an object ever be...
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
Determine whether the series converges or diverges. 00 2n + 3n n=1 15 +
-
Propose a structure for a compound C15H24O that has the following 1H NMR spectrum. The peak marked by an asterisk disappears when D2O is added to thesample. TMS 10 8. 0 ppm Chemical shift (8)...
-
The reduction of carbonyl compounds by reaction with hydride reagents (H: ? ) and the Grignard addition by reaction with organo magnesium halides (R: ? + MgBr) are examples of nucleophilic carbonyl...
-
Ethers can be prepared by reaction of an alkoxide or phenoxide ion with a primary alkyl halide. Anisole, for instance, results from reaction of sodium phenoxide with iodomethane. What kind of...
-
Employee longevity A large insurance company has developed a model to identify the factors associated with employee turnover. The dependent variable is number of years an employee stays with the...
-
5. Consider the 'top-hat' function which is zero everywhere except between -1 and 1 where it takes value 1. Show, using only elementary integration, that the convolution of this function with itself...
-
HERE IS THE SITUATION. With attendance plummeting by 50 percent in the past decade, a new, younger executive director was hired to reverse the trend. By understanding the importance of integrating...

Study smarter with the SolutionInn App


