The following data have been reported for the density, r, vs. mole fraction ethanol of binary mixtures
Question:
The following data have been reported for the density, r, vs. mole fraction ethanol of binary mixtures of ethanol (1) and formamide (2) at 25°C and 1 bar.
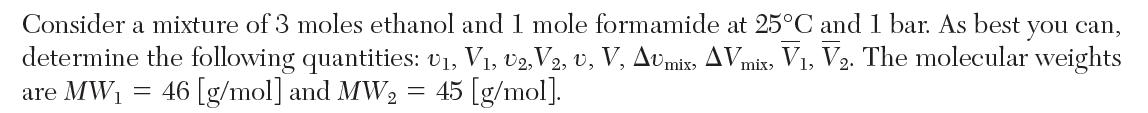
Transcribed Image Text:
X1 0 0.1000 0.1892 0.2976 0.3907 0.5009 0.5929 0.6986 0.8009 0.8995 1 p, [g/cm] 1.1314 1.0846 1.0457 1.0042 0.9678 0.9335 0.9022 0.8701 0.8401 0.8126 0.7857
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Using the information in Problems 7.13 and 8.20, estimate the heat of vaporization for the first bit of ethanol from ethanol-water solutions containing 25, 50, and 75 mol % ethanol and from a...
-
a. The following data have been reported for the vapor pressure of ethanol as a function of temperature. Use these data to calculate the heat of vaporization of ethanol at 17.33C. b. Ackermann and...
-
The following data have been reported by Shen and Smith for benzene (Bz) adsorption on silica gel [16]: a) Do these data better fit a single site or a dual site Langmuir isotherm? Why? b) Assuming...
-
How many circuits are required to accommodate kitchen appliances in an average size home?
-
Write a paper on Target Market (be sure to include demographics and psychographic analysis).Organization name is Target Dept Store. Paper should include an overview of demographics (age, income,...
-
What are the two elements that an employee must show to make a successful Title VII claim? Do you think the motivating factor standard is appropriate for discrimination claims based on national...
-
The output of a single-mode, well-stabilized laser is passed through a spatially distributed phase modulator (or a phase-only spatial light modulator that is changing with time). The field observed...
-
Paula makes the following gifts in the current year: $ 20,000 to the United Way; $ 15,000 to her brother Skip, who is a compulsive gambler; $ 45,000 to her husband Larry, to fund a new boat; and $...
-
Analyze the case analysis titled "The Cage" and answer the following questions. Explain it clearly and elaborate. 1. What should Sanjay's next steps be in preparing the GMs to take on added...
-
Consider a binary mixture of ideal gases, a and b, at temperature T and pressure P. Come up with an expression for in terms of T, P, and ya. What is the value of (AGmix) a
-
The following data are available for a binary mixture of ethanol and water at 20C: (a) Make a plot of the partial molar volumes of ethanol and water vs. mole fraction ethanol. (b) What is vmix for an...
-
Use the data in Table 1.7 and the Ketelaar triangle in Fig. 2.28 to predict the nature of the bonding in BeBr 2 , MgBr 2 , and BaBr 2 . Table 1.7. Figure 2.28. TABLE 1.7 Pauling Xp. Mulliken, XM, and...
-
In 2017, Korea's largest messenger app opened and online only bank and opened over a million accounts in just five days. What is the name of this app?
-
Consider two bonds: bond XY and bond ZW. Bond XY has a face value of $1,000 and 10 years to maturity and has just been issued at par. It bears the current market interest rate of 7% (i.e. this is the...
-
12. The following table shows risk estimates for the monthly returns on Petco stock, the Fidelity Magellan mutual fund, and the S&P 500: Asset Petco B 14.0% 1.75 Fidelity Magellan 4.4% 1.00 S&P 500...
-
Q2: Using Laplace transform find the solution of the IVP. Give all the details. a) y" + 5y +4y=54e-202, y(0) = 1 y (0) = 4 b) y" + 5y + 2y=78e - 16e*, y(0) > 1 y (0) = 7 c) y" + 3y + 5y = 69e* -...
-
On March 1, the bank agreed to lend Victoria $1,000 to buy a flat screen television by signing a security agreement. On March 3, the bank gave Victoria the money. On March 4, Victoria purchased the...
-
Document for Analysis: Poor Claim Letter Your Task. Analyze the following poorly written claim letter. List its weaknesses. If your instructor directs, revise it. Current date Mr. Morgan Monroe...
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
The FEMO theory (Problem 11.14) of conjugated molecules is rather crude and better results are obtained with simple Huckel theory. (a) For a linear conjugated polyene with each of N carbon atoms...
-
If you have access to mathematical software that can perform matrix diagonalization, use it to solve Problems 11.15 and 11.16, disregarding the expressions for the energies and coefficients given...
-
Electronic excitation of a molecule may weaken or strengthen some bonds because bonding and antibonding characteristics differ between the HOMO and the LUMO. For example, a carbon-carbon bond in a...
-
Create the following vectors in Octave a=[1 2 3]; b=[21011]; Generate the convolution of a and b: in your report explain how the first three values in the convolution were calculated.
-
Find an analytic stuxction whose reas parte. SinLy Coshly- COS2x.
-
6. The quadratic equation is y = 2x+4x-1. (a) Determine the direction of its opening and whether it is a maximum or minimum point. Explain your answer. (b) Find the vertex. (c) Find the x-intercept...

Study smarter with the SolutionInn App


