The partial molar Gibbs energy of species 1 in a binary liquid mixture of species 1 and
Question:
The partial molar Gibbs energy of species 1 in a binary liquid mixture of species 1 and 2 is given by: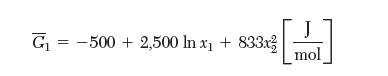

Transcribed Image Text:
G-500 + 2,500 In x + 833x J mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Romrick Albia
I am excellent student in my school. I have much interest in mathematics. I am studying from jawahar navodaya vidyalaya kaushambi and i am class topper at my school from 6 class to 12th class. I really like to clear the doubt of my classmates.
I have good skill of solving problems with easy way.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
A binary mixture of species a and b behaves as an ideal gas at 300 K and 1 bar. Calculate the partial molar Gibbs energy of species a, Ga, and the total solution Gibbs energy, g, at the following...
-
We have the following properties for a certain mixture for mixing at constant temperature and pressure: where S i , the pure-component molar entropy of component i, is given by Here S i , U i , and V...
-
At 25C and atmospheric pressure the volume change of mixing of binary liquid mixtures of species 1 and 2 is given by the equation V = x 1 x 2 (45x 1 +25x 2 ) where V is in cm 3 mol 1 . At these...
-
How does unemployment behave over the business cycle?
-
1- Describe the project manager's role in monitoring tasks. 2- Describe the project manager's role in directing resources. 3- Describe the project manager's role in managing change requests. 4-...
-
Carl Zeiss Meditec AG (Deutsche Brse XETRA: AFX), 65 percent owned by the Carl Zeiss Group, provides screening, diagnostic, and therapeutic systems for the treatment of ophthalmologic (vision)...
-
What does the variance tell us about data points?
-
On January 1, 2010, the Fastor Company had a retained earnings balance of $218,600. It is subject to a 30% corporate income tax rate. During 2010, the company earned net income of $67,000, and the...
-
Derive an expression for V = V(x,t) of the glider at the bottom of the incline plane by applying the kinematic equations of motion. x = distance traveled by glider t = time for glider to travel...
-
The following expression describes the molar volume of a binary liquid mixture of species 1 and 2: Consider a mixture that contains 2 mol of species 1 and 4 mol of species 2. Answer the following...
-
You are running a manufacturing process where a mole fraction of 0.40 species 1 in a binary mixture of 1 and 2 is required at a fl ow rate of 5 mol/s and a temperature of 300 K. This is achieved by...
-
What are settlement options? Which option should you choose?
-
Say that the hourly cost to employers per German industrial worker is $43. The hourly cost to employers per U.S. industrial worker is $39, while the average cost per Taiwanese industrial worker is...
-
Theres a gas shortage in Gasland. Youre presented with two proposals that will achieve the same level of reduction in the use of gas. Proposal A would force everybody to reduce their gas consumption...
-
If you are willing to pay $5,000 for a used car that is a cherry and $1,000 for a used car that is a lemon, how much will you be willing to offer to purchase a car if there is a 50 percent chance...
-
Suppose an air-quality law is passed that requires 3.75 percent of all the cars sold to emit zero pollution. a. What would be the likely impact of this law? b. Can you think of any way in which this...
-
In his book At the Hand of Man, Raymond Bonner argues that Africa should promote hunting, charging large fees for permits to kill animals (for example, $7,500 for a permit to shoot an elephant). a....
-
Lee Pet Supplies purchases its inventory from a variety of suppliers, some of which require a six-week lead time before delivering the goods. To ensure that she has a sufficient supply of goods on...
-
What will be the final value of DI after executing the following piece of code? Execute the instructions dependently one after another. CLD MOU CX,OFOH MOU AX.02874H MOU DI,01000H MOU ES, DI SUB...
-
An object of mass 2.0 g suspended from the end of a spring has a vibrational frequency of 3.0 Hz. Calculate the force constant of the spring.
-
Calculate the percentage difference in the fundamental vibration wave number of IH35Cl and 2H37CIon the assumption that their force constants are the same.
-
The wave number of the fundamental vibrational transition of 79BrB1Bris 323.2 cm-1, Calculate the force constant of the bond (mC9Br) = 78.9183 u, m(8IBr) = 80.9163 u).
-
Discuss employers' responsibilities for reporting workplace incidents that result in serious injury or illness. Include in your discussion the time frames for this reporting, whether or not you...
-
Which 60" aspect ratio resource is a larger screen area 4.3 or 16.9?
-
Match the items in the left column to the items in the right column. 1. check a set of written directions that tells a bank how to use money to pay someone 2. exchange costs what is given up from...

Study smarter with the SolutionInn App


