The process in Example 5.2 indicates that we need to put work into the system during an
Question:
The process in Example 5.2 indicates that we need to put work into the system during an expansion process. Determine whether this result is possible (in a thermodynamic sense); if it is, explain this result physically.
EXAMPLE 5.2
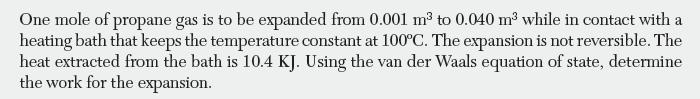
Transcribed Image Text:
One mole of propane gas is to be expanded from 0.001 m to 0.040 m while in contact with a heating bath that keeps the temperature constant at 100C. The expansion is not reversible. The heat extracted from the bath is 10.4 KJ. Using the van der Waals equation of state, determine the work for the expansion.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Let A, B be sets. Define: (a) the Cartesian product (A B) (b) the set of relations R between A and B (c) the identity relation A on the set A [3 marks] Suppose S, T are relations between A and B, and...
-
can someone solve this Modern workstations typically have memory systems that incorporate two or three levels of caching. Explain why they are designed like this. [4 marks] In order to investigate...
-
Portray in words what transforms you would have to make to your execution to some degree (a) to accomplish this and remark on the benefits and detriments of this thought.You are approached to compose...
-
Let be an arbitrary operation in Problems 5259. Describe the operation for each problem. 5038; 70 2= 9; 901 = 10; 8 0 2 = 10; -
-
Discuss the two (2) sources of innovation classified as knowledge push and need pull. Provide an example of each classification and discuss two (2) driving factors that encouraged the development of...
-
Solve Exercise 7.21 by estimating the AR parameters using the Levinson-Durbin recursions with the Burg reflection coefficients. Exercise 7.21 Use the covariance method to estimate the PSD of...
-
As sales manager, you realize your salespeople need to be more efficient and effective in managing their time and territory. As you deliver the opening comments at an all-day seminar on time and...
-
Holt Company purchased a computer for $8,000 on January 1, 2011. Straight-line depreciation is used, based on a 5-year life and a $1,000 salvage value . In 2013, the estimates are revised. Holt now...
-
The Rando Corporation's stock has a beta of 1.5. If the excess return on the stock market increases by 5%, by approximately how much will the excess return on Rando Corporation's stock increase?
-
Gas A expands through an adiabatic turbine. The inlet stream fl ows in at 100 bar and 600 K while the outlet is at 20 bar and 445 K. Calculate the work produced by the turbine. The following data are...
-
We are interested in the thermodynamic properties of a strip of rubber as it is stretched (see below). Consider n moles of pure ethylene propylene rubber (EPR) that has an unstretched length z0. If...
-
A few years ago, Tasty Food Company purchased an automatic production line at an installed cost of $325,000. The company has recognized depreciation expenses totaling $215,250 since its installation....
-
Explain the profitability index rule of thumb.
-
Why can a firm with no ongoing projects, and investment opportunities that currently have negative NPVs, still be worth a positive amount?
-
What is the difference between a cash offer and a rights offer for a seasoned equity offering?
-
Identify whether the following assets would be classified as current or non-current at the end of the reporting period. Explain your classification decision. a Cash and cash equivalents b Motor...
-
How can you decide the order of investment in a staged investment decision?
-
Internet CasePopular Reports. You have just been hired as an accountant for a large metropolitan city in the eastern part of the country. Your first assignment is to assist the finance director in...
-
Critical reading SAT scores are distributed as N(500, 100). a. Find the SAT score at the 75th percentile. b. Find the SAT score at the 25th percentile. c. Find the interquartile range for SAT scores....
-
Self-assembled monolayers (SAMs) are receiving more attention than Longmuir-Blodgett (LB) films as starting points for nanofabrication. How do SAMs differ from LB films and why are SAMs more useful...
-
Explain the physical origins of surface activity by surfactant molecules.
-
Calculate the number-average molar mass and the mass-average molar mass of a mixture of two polymers, one having M = 62 kg mol-1 and the other M = 78 kg mol-1, with their amounts (numbers of moles)...
-
Tradewind Aviation, which provides round trip flights from Teterboro airport in NJ to Martha's Vineyard and Nantucket, in Mass., recently hired a new accountant. The accountant's first task was to...
-
If a balance sheet is prepared for Cookie Creations at June 30, what balance will be reported as cash in the current assets section? If a balance sheet were prepared, cash at June 30th, 2020 would be...
-
The Chicago Men's Shop made the following purchases of dress shirts: 50 shirts at $ 78.32 in January, 60 shirts at $ 41.21 in March, 75 shirts at $ 82.74 in June, and 35 shirts at $ 61.60 in August....

Study smarter with the SolutionInn App


