Use the Rackett equation to calculate the liquid-phase molar volume of each of the following species at
Question:
Use the Rackett equation to calculate the liquid-phase molar volume of each of the following species at the same temperature as the measured values reported. Which species had the greatest absolute percent error? The least? Can the trend be explained by molecular concepts?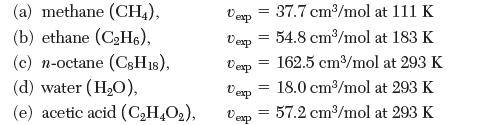
Rackett equation
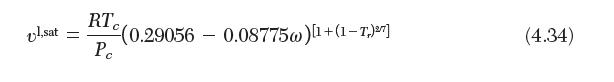
Transcribed Image Text:
(a) methane (CH4), (b) ethane (CH6), (c) n-octane (CsH18), (d) water (HO), (e) acetic acid (CH4O), Desp Vexp Vexp Dexp Dexp 37.7 cm/mol at 111 K 54.8 cm/mol at 183 K 162.5 cm/mol at 293 = 18.0 cm/mol at 293 K 57.2 cm/mol at 293 K = = = =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
(a) LEP Table 12-2: Exothermic Reaction with Heat Exchange Download the Polymath, MATLAB, Python, or Wolfram codes for the algorithm and data given in Table T12-2 for the exothermic gas phase...
-
Define change management and its significance in organizations. Discuss the challenges faced during the process of organizational change and strategies to overcome resistance.
-
Which of the items in the list of transactions in question A5.1 will have an effect on a statement of cash flows? Refer to A5.1, (a) Owner puts cash into the business. (b) Buy a vehicle for cash. (c)...
-
The following transactions apply to Walnut Enterprises for Year 1, its first year of operations: 1. Received $50,000 cash from the issue of a short-term note with a 6 percent interest rate and a...
-
At a ski resort, water at \(40{ }^{\circ} \mathrm{F}\) is pumped through a 3-in.diameter, 2000-ft-long steel pipe from a pond at an elevation of \(4286 \mathrm{ft}\) to a snow-making machine at an...
-
Determine the amount that would accumulate for the following investments: 1. $10,050 at 10% per annum, compounded annually for six years. 2. $650 at 12% per annum, compounded quarterly for 10 years....
-
Northwest Company produces two types of glass shelving: rounded edge and squared edge. The company reports the following cost data. Rounded Edge Direct materials Direct labor Overhead (using...
-
Calculate the following: (a) the volume occupied by 20 kg of ethane at 70C and 30 bar (b) the pressure needed to fi ll a 0.1 m3-vessel at room temperature to store 40 kg of ethane
-
Using the steam tables, estimate the values for the thermal expansion coeffi cient, ,and the isothermal compressibility, k, of liquid water at 20C and 100C. Symbols Used in the Steam Tables T P...
-
Joan, a single mother, has AGI of $92,000 in 2022. In September 2022, she pays $5,000 in qualified tuition for her dependent son who just started at Big University. What is Joans American Opportunity...
-
(i) What is Capital Budgeting? (ii) Why is it considered so important? (iii) It may be better to use the term capital investments, rather than capital expenditures. Do you agree? Give reasons for...
-
Suppose a stock is at $23. Interest rates are at 9%. The stock pays a dividend at a known constant rate of 3.5 percent. What stock price should we use in the Black-Scholes-Merton model for a call...
-
Your Brother Will Receive $500 A Month For The Next Eight (8) Years From An Insurance Settlement. The Interest Rate Is 6 Percent Per Year For The First Three (3) Years; And 8 Percent Per Year For The...
-
2. Write a C program to find the area of circle using functions. (Functions with input and with output) .. Area of Circle; Tr Marks 3 Input: Output:
-
Jason borrows $ 10300 for the next 4 years at a variable interest rate. Assume that the interest accumulates for 4 years at an annual nominal rate of 11% compounded quarterly. Calculate the...
-
Financial statements in the United States rely heavily on historical cost information, particularly in the valuation of land, buildings, and equipment. However, International Accounting Standards...
-
A consumer magazine is evaluating five brands of trash compactors for their effectiveness in reducing the volume of typical household products that are discarded. In the experiment, each block...
-
From among the compounds chlorobenzene, o-dichlorobenzene, and p-dichlorobenzene, choose the one that (a) Gives the simplest 1H NMR spectrum (b) Gives the simplest 13C NMR spectrum (c) Has three...
-
Compounds A and B are isomers of molecular formula C10H14. Identify each one on the basis of the 13C NMR spectra presented in Figure 13.41.
-
A compound (C8H10O) has the infrared and 1H NMR spectra presented in Figure 13.42. What is its structure?
-
ABC Inc. has been in business for the past century. It requires the service of a reliable machine needed for an existing line of business that has been very successful over the past few decades. ABC...
-
A piston-cylinder assembly contains 1.00 mole of ideal diatomic gas initially at 500 kPa and 280 K. Maintaining constant temperature the piston pressure is reduced instantly to 200 kPa. What is the...
-
Sales promotions At the bottom of all its February sales receipts, Seifert Stores Inc. printed $2-off coupons. The coupons may be redeemed March 1-April 30. Seifert Stores Inc. accepts only cash,...

Study smarter with the SolutionInn App


