Using the steam tables, estimate the values for the thermal expansion coeffi cient, ,and the isothermal compressibility,
Question:
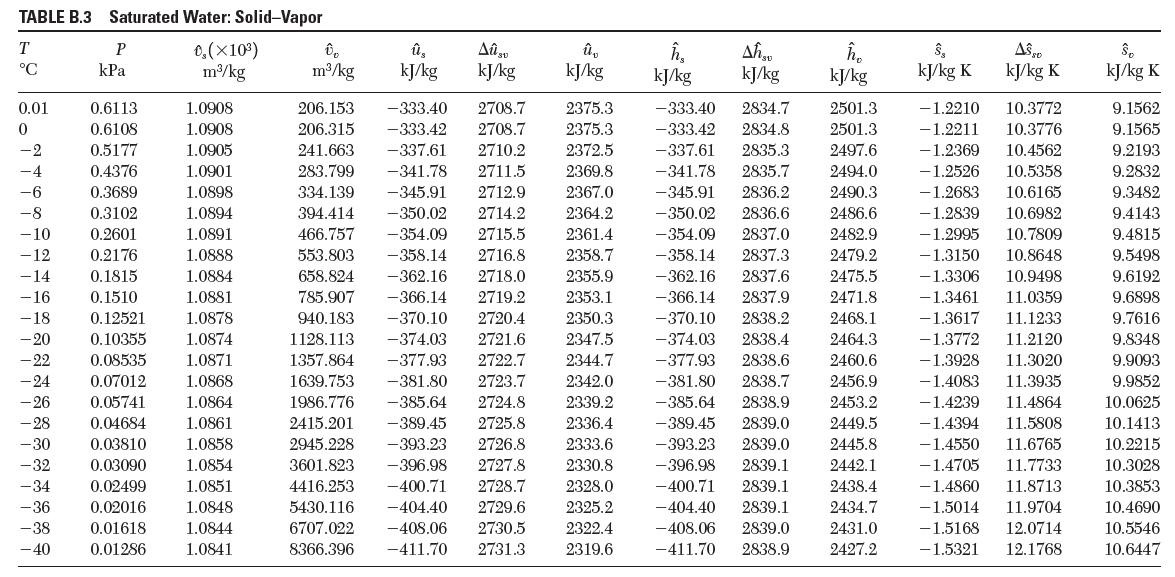
Using the steam tables, estimate the values for the thermal expansion coeffi cient, β,and the isothermal compressibility, k, of liquid water at 20°C and 100°C.
Transcribed Image Text:
Symbols Used in the Steam Tables T P Subscripts 1 S V lv su Temperature Pressure Specific volume Specific internal energy Specific enthalpy Specific entropy C kPa or MPa m/kg kJ/kg kJ/kg kJ/kg K Liquid in equilibrium with vapor Solid in equilibrium with vapor Vapor in equilibrium with liquid or solid Change by evaporation Change by sublimation
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
what ways can organizations leverage strategic partnerships, alliances, and ecosystem collaborations to enhance strategic planning outcomes, leverage complementary capabilities, and exploit synergies...
-
Shalit company uses job order cost system and has established a predetermined overhead application rate for the current year of 150% of direct labor cost,based on budgeted overhead of $1800000 and...
-
This question involves using the steam tables in Appendix A, but answer questions A through C before looking at the steam tables. A . The density of liquid water at ambient conditions is about 1 g/cm...
-
Internal Company Assessment (Strategic Analysis Output) on Riise Ev converting Canada post delivery cars to electronic finding out firm strengths weakness liabilities problem constraints...
-
Which of the items in the list of transactions in question A5.1 will have an effect on an income statement (profit and loss account)? (a) Owner puts cash into the business. (b) Buy a vehicle for...
-
Colson Company has a line of credit with Federal Bank. Colson can borrow up to $800,000 at any time over the course of the Year 1 calendar year. The following table shows the prime rate expressed as...
-
Water at \(10{ }^{\circ} \mathrm{C}\) is pumped from a lake as shown in Fig. P8.72. If the flowrate is \(0.011 \mathrm{~m}^{3} / \mathrm{s}\), what is the maximum length inlet pipe, \(\ell\), that...
-
1. Can Mary set up an IRA and deduct the contribution from her income that is subject to federal income taxation? Does the same apply to Jason? Could Marys or Jasons children have IRA accounts? 2....
-
Here are selected data for Propel Corporation: Beginning raw materials inventory $37,000 Beginning work in process inventory $61,500 Beginning finished goods inventory $57,100 Cost of materials...
-
Use the Rackett equation to calculate the liquid-phase molar volume of each of the following species at the same temperature as the measured values reported. Which species had the greatest absolute...
-
Determine expressions for the thermal expansion coeffi cient, , and the isothermal compressibility, k, for an ideal gas.
-
A diamond-shaped loop of wire is pulled at a constant velocity through a region where the magnetic field is directed into the paper in the left half and is zero in the right half, as shown in Figure...
-
on January 1, 2000, the price of koka kola was $10. on jan and, 2020, the shares were worth $100. the stock Paid no dividends during the period. what is the annual geometric return.
-
Billy and Sally just won $105,000 in the California Lottery. They spent $30,000 on a new Lexus vehicle, and invest the rest in an investment earning 4.75% compounded annually. They will sell the...
-
Find the volume of the hexagonal prism whose base is a regular hexagon of side x = 10cm.
-
What are three actions a manager can take to ensure they are providing motivational "health food" to your employees. Optimal Motivation Susan Fowler When it comes to motivation, the reason you are...
-
The average variable cost of producing 240 units is $0.32. What is the present value of 8 years of annual cash receipts of $19,200 at the end of each year that begins two years from today, assuming a...
-
Conserv Corporation, a computer software company, is trying to determine the appropriate accounting procedure to apply to its software development costs. Management is considering capitalizing the...
-
Chao, Louis, and Mari, unrelated individuals, own all of the shares of Cerise Corporation. All three shareholders have been active in the management of Cerise since its inception. In the current...
-
Deduce the structure of a compound having the mass spectrum and 1H NMR spectrum presented in Figure 13.43.
-
Figure 13.44 presents several types of spectroscopic data (IR, 1H NMR, 13C NMR, and mass spectra) for a particular compound. What is it?
-
[18]-Annulene exhibits a 1H NMR spectrum that is unusual in that in addition to a peak at ? 8.8 ppm, it contains a second peak having a chemical shift ? of - 1.9 ppm. A negative value for the...
-
Review of Uniqlo's Communication of Ethics and Approach to CSR and Sustainability in Relation of Ethical Marketing Strategies.
-
The costs per equivalent unit of direct materials and conversion in the Rolling Department of Jabari Steel Company are $375 and $60, respectively. The equivalent units to be assigned costs are as...
-
A Rankine cycle steam power plant operates at a boiler pressure of 2.0 MPa and a condenser pressure of 0.100 MPa. The superheat temperature is 250 o C. Using the steam tables how much work is...

Study smarter with the SolutionInn App


