Using data from the steam tables, come up with an expression for the ideal gas heat capacity
Question:
Using data from the steam tables, come up with an expression for the ideal gas heat capacity of H2O in the form:![]()
Compare your answer to the values in Appendix A.2.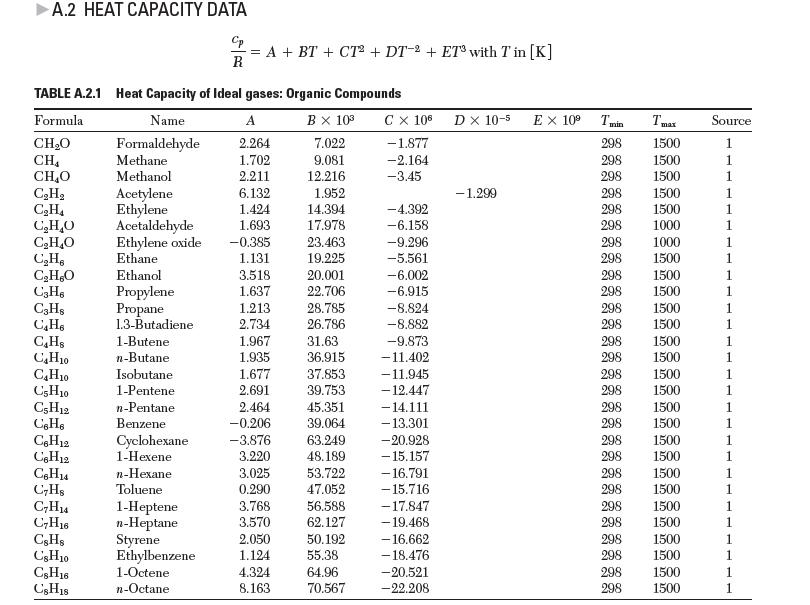
Transcribed Image Text:
Co C = A + BT
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Bree Normandin
Success in writing necessitates a commitment to grammatical excellence, a profound knack to pursue information, and a staunch adherence to deadlines, and the requirements of the individual publication. My background comprises writing research projects, research meta-analyses, literature reviews, white paper reports, multimedia projects, reports for peer-reviewed journals, among others. I work efficiently, with ease and deliver high-quality outputs within the stipulated deadline. I am proficient in APA, MLA, and Harvard referencing styles. I have good taste in writing and reading. I understand that this is a long standing and coupled with excellent research skills, analysis, well-articulated expressions, teamwork, availability all summed up by patience and passion. I put primacy on client satisfaction to gain loyalty, and trust for future projects. As a detail-oriented researcher with extensive experience surpassing eight years crafting high-quality custom written essays and numerous academic publications, I am confident that I could considerably exceed your expectations for the role of a freelance academic writer.
5.00+
7+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
What are the expected readings of the ammeter and voltmeter for the circuit inFigure? 6.00 2 10.0 2 6.00 V 5.00 2 4.50 V 6.00 2
-
In a television weather report a "wind chill factor" is frequently stated. The actual factor is based on empirical data. You are asked to come up with an expression for wind chill based on the...
-
One kilogram of steam is placed in a piston-cylinder device, initially at P = 1 bar and T = 100C. The steam is heated at constant pressure to T = 200C. A . Find the heat and work for this process....
-
The combined sewer system in city ABC is comprised of two parallel interceptors referred to as "North" and "South" lines. The southern line is connected to a newly built wastewater treatment plant....
-
Explain why stocks traded on the NYSE generally exhibit less risk than stocks that are traded on other exchanges.
-
Assume that the 6 spoiled units of Textual Brilliances Job #10 can be reworked for a total cost of $1,800. A total cost of $6,600 associated with these units has already been assigned to Job #10...
-
Describe common legal issues for behavioral health professionals.
-
In 2011, Alliant Corporation acquired Centerpoint Inc. for $300 million, of which $50 million was allocated to goodwill . At the end of 2013, management has provided the following information for a...
-
Cullumber Company started business on January 1, 2024. Some of the events that occurred in its first year of operations follow: Transactions 1. Equipment that cost $208,400 was purchased on February...
-
Steam at 8 MPa and 500C fl ows through a throttling device, where it exits at 100 kPa. What is the exit temperature?
-
A mass fl ow controller (MFC) is used to accurately control the molar fl ow rate of gases into a system. A schematic of an MFC is shown below. It consists of a main tube and a sensor tube, to which a...
-
In Exercises 39 through 46, find the largest and smallest values of the given function over the prescribed closed, bounded interval. h(s) = 2s ln s s 2 for 0.5 s 2
-
Your team has been asked to test and document enhancements to a web application that allows buyers to purchase custom-printed canvas shoes. The tasks and dependencies are as follows: a testing plan...
-
The total market value of your portfolio is $20,000,000 and the beta of the portfolio is 1.6. It is 3/23 and you want to hedge till 5/15. You have the following contracts on the S&P500 index...
-
1. [10 Points] Consider the following context-free grammar G: S aSaSbbb | a (a) (2 points) Describe L(G). (b) (3 points) Show that G is an ambiguous grammar. (c) (5 points) Give an unambiguous...
-
Consider the HTML Cats Dogs Fish Elephants Write a CSS rule set that puts 10 pixels of spac around each column in the table.
-
9.31 A jet transport aircraft has two turbofan engines and the following characteristics: mro = 60,000 kg, S = 150 m; TL =2 x 80 kN, b= 35 m; e=0.82; Cpo=0.02, CLmax = 2.6. a. Determine the corner...
-
What relationship exists between the size of the standard deviation and the degree of asset risk?
-
Sundial Technologies produces and sells customized network systems in New Brunswick. The company offers a 60-day, all software and labor-and an extra 90-day, parts-only- warranty on all of its...
-
Like hydrogen peroxide, the inorganic substances hydrazine (H2NNH2) and hydroxylamine (H2NOH) possess conformational mobility. Write structural representations or build molecular models of two...
-
Of the three conformations of propane shown, which one is the most stable? Which one is the least stable? Why?
-
Sight down the C-2C-3 bond, and draw Newman projection formulas for the (a) Most stable conformation of 2, 2-dimethylbutane (b) Two most stable conformations of 2-methylbutane (c) Two most stable...
-
Why would someone place "Patent Pending" on their product if they had not received a patent for it?
-
Explain the graph below. Talk about market risk, firm-specific risk, diversification and number of stocks in a portfolio Does that mean you need to buy at least 20 stocks? What are the possible...
-
Why would "Discovery" play a role in causing a plaintiff or defendant to seek an alternative to going to court?

Study smarter with the SolutionInn App


