You need to fi nd the triple point of pure species A and are unable to fi
Question:
You need to fi nd the triple point of pure species A and are unable to fi nd its value in any reference books. You can fi nd the following data for A. It sublimes at 200 K and 0.1 bar, and it boils at 250 K and 1 bar.
(a) Calculate the temperature and pressure at the triple point. You may assume the enthalpies of sublimation and vaporization are constant. The values are: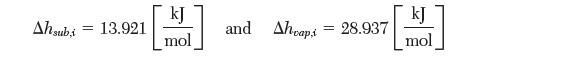
(b) It turns out that the enthalpy changes are better represented in the following forms:![]()
where A, B, C, and D are constants. Come up with an expression to fi nd T and P at the triple point with these forms.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





