You wish to determine the fugacity of a in a liquid mixture with 1 mole of a
Question:
You wish to determine the fugacity of a in a liquid mixture with 1 mole of a and 4 moles of b at 30 kPa and 20°C. At this temperature, the saturation pressure of pure a is 50 kPa. The excess Gibbs energy for a mixture of a and b has been fi t to the following relation: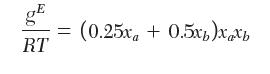
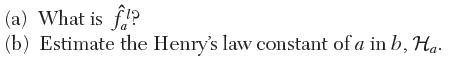
Transcribed Image Text:
gE = (0.25x + 0.5x)x a Xb
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Madhvendra Pandey
Hi! I am Madhvendra, and I am your new friend ready to help you in the field of business, accounting, and finance. I am a College graduate in B.Com, and currently pursuing a Chartered Accountancy course (i.e equivalent to CPA in the USA). I have around 3 years of experience in the field of Financial Accounts, finance and, business studies, thereby looking forward to sharing those experiences in such a way that finds suitable solutions to your query.
Thus, please feel free to contact me regarding the same.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
You wish to determine the sequence of a polypeptide that has the following amino acid composition. (a) What is the maximum number of peptides you can expect if you cleave the polypeptide with...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Suppose the (1) + (2) system exhibits liquid-liquid immiscibility. Suppose we are at a state where G 1 /RT = 0.1 and G 2 /RT = 0.3. The Gibbs energy of mixing quantifies the Gibbs energy of the...
-
The following are the Ledger Balance (in thousands) extracted from the books of Vaishnavi Bank Ltd as on March 31, 2016. The bank's Profit and Loss Account for the year ended and Balance Sheet as at...
-
Wonder Kid Enterprises Company has produced the following results over the last three years: Management has just issued its annual report for 20XZ and a fair reading of their commentary is that the...
-
The height of a radio tower is 500 feet, and the ground on one side of the tower slopes upward at an angle of 10 (see the figure). (a) How long should a guy wire be if it is to connect to the top of...
-
The \(45^{\circ}\) off-axis test shown in Figure 10.37 is conducted on a 10 in. \(\times 1\) in. \(\times 0.1\) in. \((254 \mathrm{~mm} \times 25.4 \mathrm{~mm} \times 2.54 \mathrm{~mm})\)...
-
Air France-KLM (AF), a Franco-Dutch company, prepares its financial statements according to International Financial Reporting Standards. AF's financial statements and disclosure notes for the year...
-
Determine the cost of a product with a selling price of $45.55, ober head of 5.50$ and net profit of 9$.
-
Calculate the activity coeffi cients of a binary mixture of 30 mol% acetone (1) in water (2) at 61.1C using the UNIQUAC activity coeffi cient model. Compare the values to the experimentally measured...
-
Your supervisor has assigned you to obtain parameters A and B for the three-suffi x Margules equation to input in the companys phase equilibrium computer database. The binary mixture of interest is...
-
Draw the K-maps of these sum-of-products expansions in three variables. a) x b) yz + c) xyz + xy + y + z
-
Company 1 Company 2 Company 3 EBIT interest coverage 16.7 2.7 12.8 EBITDA interest coverage 24.6 3.7 18.7 FFO/ Total debt 135.1 19.8 80.2 Free operating cash flow/Total debt 87.9 8.2 40.6 Total...
-
Read the following statements and answer whether TRUE or FALSE. Provide a reason for your answer. Q.1.1 Q.1.2 Q.1.3 Q.1.4 Q.1.5 Q.1.6 Q.1.7 Q.1.8 Q.1.9 In the context of the legal profession, ethics...
-
At December 31, 2023, Bonita Corporation has the following account balances: Bonds payable, due January 1, 2033 Interest payable $1,933,000 85,000 Show how the above accounts should be presented on...
-
Briefly describe what cultural and cross-cultural training can contribute toward employees' attitudes and behaviors in diverse workplaces.
-
Assuming monetary benefits of an information system at $100,000 per year starting in year 1, one-time costs of $85,000 in year 0, recurring costs of $25,000 per year starting in year 1, a discount...
-
In a small group, conduct a SWOT analysis, develop objectives, and create a marketing strategy for your school, a student organization you might be involved in, or a local business.
-
Discrete sample spaces: suppose there are N cable cars in San Francisco, numbered sequentially from 1 to N. You see a cable car at random; it is numbered 203. You wish to estimate N. (See Goodman,...
-
In an experiment to measure the molar mass of a gas, 250 cm3 of the gas was confined in a glass vessel. The pressure was 152 Torr at 298 K and, after correcting for buoyancy effects, the mass of the...
-
A certain sample of a gas has a volume of20.00 dm ' at OCand 1.000 atm. A plot of the experimental data of its volume against the Celsius temperature, , at constant p, gives a straight line of slope...
-
A certain gas obeys the van der Waals equation with a =0.76 m6 Pa mol-2, its volume is found to be 4.00 X 10-4 m3 mol-1 at 288 K and 4.0 MPa. From this information calculate the van der Waals...
-
analyze the attached Seagate case. In your analysis, you should address the following questions: 1) Why is Seagate undertaking this transaction? Is it necessary to divest the Veritas shares in a...
-
What is the most essential attribute of an effective key risk indicator (KRI)? Discuss them in details and give example of each.
-
READ ATTACHED LINK TO ANSWER THE FOLLOWING QUESTIONS: (copy and paste link for it to work!) Questions: 4. Identify the Needs, Priorities, and Concerns of the family and the child. 5. Identify...

Study smarter with the SolutionInn App


