Calculate the activity coeffi cients of a binary mixture of 30 mol% acetone (1) in water (2)
Question:
Calculate the activity coeffi cients of a binary mixture of 30 mol% acetone (1) in water (2) at 61.1°C using the UNIQUAC activity coeffi cient model. Compare the values to the experimentally measured values, ![]() The UNIQUAC parameters are reported as follows:
The UNIQUAC parameters are reported as follows: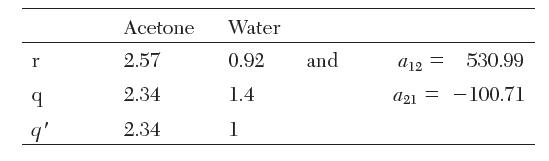
Transcribed Image Text:
exp = = 2.30 and 1 YExp exp = 1.32.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)

Answered By

Stacy kosgei
I offer quality, original and timely services; Highly credible and void of plagiarism. Your success is my pleasure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Calculate the activity coeffi cients of a binary mixture of 20.0 mol% acetone (1) in chloroform (2) at 35.17C using the UNIQUAC activity coeffi cient model. The UNIQUAC parameters are reported as...
-
Calculate the activity coeffi cients of a binary mixture of 41.5 mol% ethanol (1) in benzene (2) at 45C using the UNIQUAC activity coeffi cient model. The UNIQUAC parameters are reported as follows:...
-
Prepare a plot of the activity coeffi cients of a binary mixture of ethanol (1) and n-heptane (2) versus ethanol mole fraction in a binary mixture at 50C using the UNIQUAC activity coeffi cient...
-
State whether or not each of the following events would result in a liability being recognised in the accounts at 30 June. 1. Taxes for the year ended 30 June, which are not payable until October. 2....
-
From the following account balances for June 2016, prepare in proper form (a) An income statement, (b) A statement of owner's equity, and (c) A balance sheet for French Realty. $3,310 S. French,...
-
In attempting to fly from Chicago to Louisville, a distance of 330 miles, a pilot inadvertently took a course that was 10 in error, as indicated in the figure. (a) If the aircraft maintains an...
-
A \(45^{\circ}\) off-axis specimen cut from an AS/3501 carbon/epoxy lamina is subjected to a tensile test. The specimen is \(3 \mathrm{~mm}\) thick and \(25 \mathrm{~mm}\) wide, and a tensile load of...
-
IT auditors need people skills as well as technical skills. One such skill is the ability to interview effectively. Discuss some techniques or tools that might help an interviewer get the best...
-
Bostian, Inc. has total assets of $540,000. Its total debt outstanding is $185,000. The Board of Directors has directed the CFO to move towards a debtto-assets ratio of 55%. How much debt must the...
-
Wilson parameters for mixtures of ethanol (1), 1-propanol (2), and water (3) at 60C are reported as follows: Calculate the fugacity of ethanol in a liquid mixture containing 30% ethanol, 20%...
-
You wish to determine the fugacity of a in a liquid mixture with 1 mole of a and 4 moles of b at 30 kPa and 20C. At this temperature, the saturation pressure of pure a is 50 kPa. The excess Gibbs...
-
Your organization's first Web site was launched just six months ago, but already management is calling it a complete disaster. The site has failed to stimulate additional sales and has proven to be...
-
Income and expenses information John and Mary Smith Annual Income John Salary $230,000 Mary Salary $280,000 BP Stock Option Bonus (ISO) Family Business Income $75,000 $ 100,000 2023 Tax Liability...
-
Chef Grill Company manufactures two types of cooking grills: the Gas Cooker and the Charcoal Smoker. The Cooker is a premium product sold in upscale outdoor shops; the Smoker is sold in major...
-
"Would it be possible for me, SFC Nanola, Ed V. 140-04-7125, to qualify for MILPER Number 24-012 Temporary Change to Timeline for Requesting Voluntary Retirement from Active Federal Service?...
-
For the next three questions, please use the Newman and Case Corp. financial statements provided below. and Corp 2015 Income Se Da Cost of Good S Oher Exper prector, FERT How Toute rume Tom Netcome...
-
2019 April 1 Purchased Land, building and equipment for $320,000 by signing a note payable. The appraised value for the new assets is Land - $138,000, Building - $276,000, Equipment - $46,000. The...
-
Explain the role of a chief marketing officer. Summarize an article that describes the importance of this position, the characteristics of an effective officer, or any issues surrounding this...
-
You are a U.S. investor who purchased British securities for 2,000 one year ago when the British pound cost U.S. $1.50. What is your total return (based on U.S. dollars) if the value of the...
-
At 100C and 1.60 kPa, the mass density of phosphorus vapour is 0.6388 kg m-3. What is the molecular formula of phosphorus under these conditions?
-
Calculate the mass of water vapour present in a room of volume 250 m3 that contains air at 23C on a day when the relative humidity is 53 per cent.
-
A gas mixture consists of 320 mg of methane, 175 mg of argon, and 225 mg of neon. The partial pressure of neon at 300 K is 8.87 kPa. Calculate (a) The volume and (b) The total pressure of the mixture.
-
a. Study the following java code snippet. public class Doctor extends HospitalStaff { private String deptName; // doctor's department name public void read Body Temperature( ) { } System.out.println(...
-
Study the code below and use it to answer the questions that follow: public class Clinic extends Hospital { public static int numOfBeds; public int numberOfPatients ; public String wardName; public...
-
Find the bandwidth for the signal: a)[(4/)[sin(2ft) + (1/3)sin(2(3f)t) + (1/5)sin(2(5f)t) +(1/7)sin(2(7f)t)] b) S(t)=sin(2ft)+sin(2(3f)t) c) a signal that ranges from 40 KHz to 4 MHz?

Study smarter with the SolutionInn App


