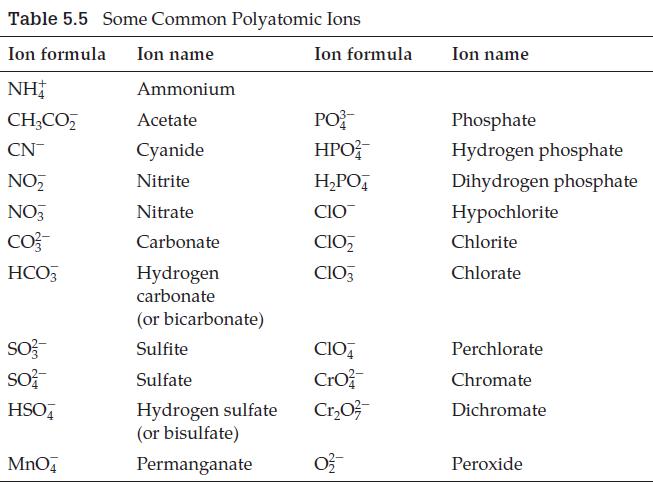
A polyatomic ion not listed in Table 5.5 is iodate, IO 3 . What is the
Question:
A polyatomic ion not listed in Table 5.5 is iodate, IO3–. What is the formula for the periodate ion, and what is the formula for magnesium periodate?
Transcribed Image Text:
Table 5.5 Some Common Polyatomic Ions Ion formula Ion name NH Ammonium CH3CO₂ Acetate CN Cyanide NO₂ Nitrite NO3 Nitrate CO²- Carbonate HCO3 Hydrogen carbonate SO3- SO² HSO4 MnO4 (or bicarbonate) Sulfite Sulfate Hydrogen sulfate (or bisulfate) Permanganate Ion formula PO HPO²/ H₂PO4 CIO CIO₂ CIO3 CIO4 Cro Cr₂0²/ 0²2/ Ion name Phosphate Hydrogen phosphate Dihydrogen phosphate Hypochlorite Chlorite Chlorate Perchlorate Chromate Dichromate Peroxide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The formula for the periodate ion is IO4 The periodate i...View the full answer

Answered By

Michael Owens
I am a competent Software Engineer with sufficient experience in web applications development using the following programming languages:-
HTML5, CSS3, PHP, JAVASCRIPT, TYPESCRIPT AND SQL.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Examine the processes in Example 13.2 One of the processes that was rejected in the second round of decision making has great potential for making the integral bladed hub for the fan from an aluminum...
-
The iodate ion is reduced by sulfite according to the following reaction: The rate of this reaction is found to be first order in IO3-, first order in SO32-, and first order in H+. (a) Write the rate...
-
Let A = [a;l E Fxn be a square n x n matrix. We define the trace of A to be the number Tr(A) = akk- k=1 a) Prove that Tr : FXn F is a linear function. b) Let A, B e F"X". Prove that Tr(AB) = Tr(BA)....
-
DoorDash acquired 51% of the common stock of Wolt on December 31, 2021, for $8,100,000,000. At the date of acquisition, Wolt reported common stock with a par value of $10,000,000,000, additional...
-
In tabular form, compare the total materials cost transferred to Work in Process and the cost of the ending inventory for each method used in E2-6, E2-7, and E2-8. Discuss the effect that each method...
-
After watching the video on outsourcing, discuss the following: 1. What role does the sourcing agent play in the 4Ps of marketing? 2. How does the outsourcing approach challenge the requirements of...
-
On March 13, 2009, Juan Mendez Sr. was admitted to a nursing facility. On that day, a doctor employed by the facility determined the father lacked the capacity to give informed consent or make...
-
Lahey Corp. has three defined-benefit pension plans as follows. How will Lahey report these multiple plans in its financial statements? Plan X Plan Y Plan Z Pension Assets (at Fair Value) $600,000...
-
(1) Write a MATLAB script to find area using the Simpson's 3/8 rule. The code should perform all checks on the number of data points required for application of the rule assuming equally spaced data....
-
The oxide ion is O 2 . How does this ion differ from the peroxide ion, O 2 2 ? Draw dot diagrams for both.
-
Consider the sulfate, sulfite, nitrate, nitrite, chlorate, and chlorite ions. What information do the -ate and -ite suffixes communicate?
-
Mark and Lisa were divorced in 2013. In 2014, Lisa has custody of their children, but Mark provides nearly all of their support. Who is entitled to claim the children as dependents?
-
Considerable evidence exists that supportive managerial and supervisor behaviours in environment initiatives are important in developing employee engagement in environmental management (EM). For...
-
NSG is a recently created UK work organisation, consisting of two operating companies Cook and Dickens Services who enjoy an equal partnership while continuing to retain separate identities and...
-
You have read the first chapter of this textbook and have taken over a company that you now discover is losing 100,000 a week. At the rate things are going, the company will not have any cash left in...
-
Many companies have a set of appraisal methods that they recommend their managers use when considering new project investments. Assume that your company uses three methods: NPV (hurdle rate of 20 per...
-
Choose any company from your country and develop a chronology of the firms life cycle, from the business start-up, through initial expansion, to stock exchange listing and then (if applicable) death...
-
Express Detail, Inc. provides mobile car washing and detailing to its customers. The Income Statement for the month ended January 31, 2012, the Balance Sheet for December 31, 2011, and details of...
-
Separate variables and use partial fractions to solve the initial value problems in Problems 18. Use either the exact solution or a computer-generated slope field to sketch the graphs of several...
-
Aspartame (below) is an artificial sweetener used in diet soft drinks and is marketed under many trade names, including Equal TM and Nutrasweet TM . In the body, aspartame is hydrolyzed to produce...
-
Draw a plausible mechanism for each of the following transformations: a. b. c. d. e. Pyridine CI
-
Ethyl trichloroacetate is significantly more reactive toward hydrolysis than ethyl acetate. Explain this observation.
-
Evaluate Hugos' list of "Strategic System Builders" (p. 268) relative to identifying and pursuing supply chain (SC) opportunities. What is the value/effectiveness of these guidelines? What are the...
-
Describe the shape and factors that affect the demand curve and the supply curve in a market. Discuss the concept of market equilibrium and how changes in demand and supply affect the equilibrium...
-
Why is important that evaluation should begin during planning and implementation and continue through the completion of the project, and how this benefits the proposed project.

Study smarter with the SolutionInn App


