Calculate the value of K eq for the NO reaction, given the following data: Start Reactants N(g)
Question:
Calculate the value of Keq for the NO reaction, given the following data:
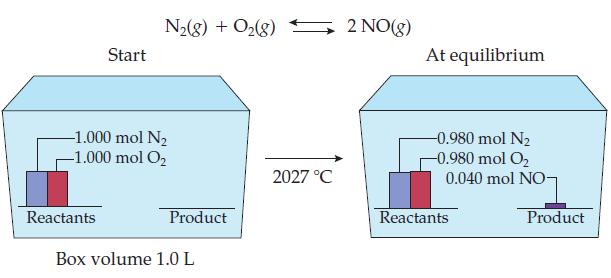
Transcribed Image Text:
Start Reactants N₂(g) + O₂(g) 2 NO(g) -1.000 mol N₂ -1.000 mol O₂ Product Box volume 1.0 L 2027 °C At equilibrium -0.980 mol N₂ -0.980 mol O₂ 0.040 mol NO- Reactants Product
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Keq NO ...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
We sell 5 0 . 0 0 0 units of a special material using 5 % of the capacity. There is a new order consisting 3 0 . 0 0 0 units from a foreign market for 6 0 0 $ . The managers of the business suggested...
-
QUESTION 1 Woolworths Ltd sells new shares to institutional investors. (a) Is this an investing, financing or dividend decision by Woolworths? (b) Is this an investing, financing or dividend decision...
-
For the reaction C2H2(g) + 4F2(g) 2CF4(g) + H2(g) So is equal to -358 J/ K. Use this value and data from Appendix 4 to calculate the value of So for CF4(g).
-
A 50 coaxial cable is connected to an 80 resistive load and a dc source with zero internal resistance. Calculate the voltage reflection coefficients at the source and at the load.
-
Cost management with an ABC system Hayne Corporation manufactures two different coffee makers, Professional for commercial use and Home for family use. Dwight Hayne, the president, recently received...
-
Should a project be accepted or rejected if the project has an internal rate of return that is less than the companys hurdle rate? You are the CFO for an automobile manufacturer. The board of...
-
A wire connected between the terminals of a battery carries a current. The wire is removed and stretched, decreasing its cross-section area and increasing its length. When the wire is reconnected to...
-
You and several friends are about to prepare a lasagna dinner. The tasks to be performed, their immediate predecessors, and their estimated durations are as follows: *There is none in the...
-
Laker Company reported the following January purchases and sales data for its only product. For specific identification. ending inventory consists of 180 units from the January 30 purchase, 5 units...
-
Consider the nuclear process shown below. (a) Identify the atoms on the left (the reactant atoms). (b) Is this fission, fusion or decay? Explain. (c) Would this process take a little or a lot of...
-
A reaction starting with just reactants will eventually reach equilibrium because: (a) The rates of the forward and reverse reactions will remain constant as the reaction proceeds. (b) The forward...
-
A sloped cathedral ceiling is between walls that are 7.50 ft high and 12.0 ft high. If the walls are 15.0 ft apart, at what angle does the ceiling rise?
-
Use the Divergence Theorem to evaluate the flux \(\iint_{\mathcal{S}} \mathbf{F} \cdot d \mathbf{S}\). \(\mathbf{F}(x, y, z)=\langle x+y, z, z-xangle, \mathcal{S}\) is the boundary of the region...
-
You are given the following information: Determine: (a) the interest rate implicit in the lease; (b) the finance charge to be allocated in each accounting period using the actuarial method; (c) the...
-
For any healthcare activity, four performance factors can be measured: structure, process, outcome, and patient experience. Identify one measure from each of these categories that could be used to...
-
Let \(\mathbf{F}\) and \(\mathbf{G}\) be vector fields. Prove the the following Product Rule for Divergence: \[ \operatorname{div}(\mathbf{F} \times \mathbf{G})=\operatorname{curl}(\mathbf{F}) \cdot...
-
\(\Delta\) denotes the Laplace operator defined by Prove the identity \[ \operatorname{curl}(\operatorname{curl}(\mathbf{F}))=abla(\operatorname{div}(\mathbf{F}))-\Delta \mathbf{F} \] where \(\Delta...
-
Explain the following aspects of the Equal Pay Act: a. Its purpose. b. The remedies available under the act. c. The defenses available to employers.
-
The population of Detroit, Michigan, decreased from 1,027,974 in 1990 to 688,701 in 2013 (Source: U.S. Census Bureau). Find the average rate of change in the population of Detroit, Michigan, over the...
-
A branch voltage in an RLC circuit is described by If the initial conditions are v(0) = 0 = dv(0)/dt, find v(t). t'v+4dv + 8v= 120 df dt
-
The step response of an RLC circuit is given by Given that i(0) = 18 A and di(0)/dt = 36 A/s, solve for i(t). di 2al + 5i = 30 dt d
-
Design a parallel RLC circuit that has the characteristic equation s 2 + 100s + 10 6 = 0.
-
Presented below are the ending balances of accounts for the Kansas Instruments Corporation at December 31, 2024. Account Title Cash Accounts receivable Raw materials Notes receivable Interest...
-
You have a goal of building an emergency account. You open the account with $500 and have a goal to build it to 12 months worth of living expenses. You figured you could live for one year off...
-
Pearce's Cricket Farm issued a 20-year, 10% semiannual bond 4 years ago. The bond currently sells for 97% of its face value. The company's tax rate is 38%. Suppose the book value of the debt issue is...

Study smarter with the SolutionInn App


