Ethane can be converted into an aldehyde and an alcohol (shown below): (a) Explain why this is
Question:
Ethane can be converted into an aldehyde and an alcohol (shown below):
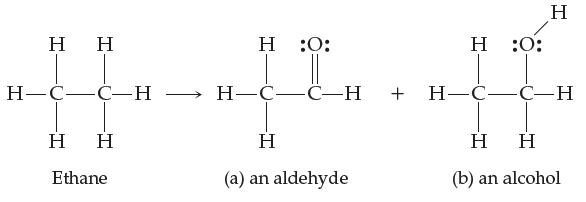
(a) Explain why this is an example of oxidation.
(b) Which is the more oxidized form of ethane, the aldehyde or the alcohol?
Transcribed Image Text:
Η Η H=C=C-H Η Η Ethane Η :O: Η Η (a) an aldehyde Τ :O: Η H-C-C-H + H-C-C-H Η Η (b) an alcohol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a The conversion of ethane into an aldehyde and an alcohol involves the process of oxidation Oxidati...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
There are two options to buy a plot of land for construction: (i) upfront payment of $200 000, or (ii) four equal payments of $50 000 in years 1-4 and the fifth payment of $100 000 in year 5. What is...
-
The conversion of natural gas, which is mostly methane, into products that contain two or more carbon atoms, such as ethane (C2H6), is a very important industrial chemical process. In principle,...
-
Read the following article and answer the questions below: Building Competitive Advantage Through People Magazine: Winter 2002Research Feature January 15, 2002 Reading Time: 23 min Christopher A....
-
William made the following acquisitions of preference shares in Pangol plc: In January 2020, the company made a 1 for 4 bonus issue. In February 2021, William sold 450 shares for 4 per share....
-
How do you combine two packaged food companies, both with very well known household brand names, and make it work? Thats the challenge managers at General Mills faced when it acquired Pillsbury. So...
-
An accountant with the Atlanta Olympic Games was charged with embezzling over $60,000 to purchase a Mercedes-Benz and to invest in a certificate of deposit. Police alleged that he created fictitious...
-
Why is it advisable to obtain a written confession from the subject of an investigation?
-
Record the following transactions of the State of Delaware general government activities for the year ended June 30, 20X6. (Amounts are stated in thousands of dollars.) Make all required entries. 1....
-
Mr. Amar is 30 year old, newly married and a successful actor in the Indian film industry. Right from his struggling days, Amar always saved a part of his income and invested in safe instruments like...
-
(a) Use the following results to arrange the four metals, W, X, Y, Z in the vertical column on the right from the most active (at the top) to least active (on the bottom). (b) Based on your series in...
-
Most combustible materials will burn in chlorine (Cl 2 ) gas. For example, methane burns to yield CH 3 Cl, CH 2 Cl 2 , CHCl 3 , and CCl 4 . Why is the word burn used in this case?
-
Use the change-of-base rule (with either common or natural logarithms) to approximate each logarithm to four decimal places. log 3 12
-
What is the Huffman coding algorithm used for? Specifying the basic principles of the algorithm, Explain in detail on a small example.
-
Find the slope of the tangent line at x = 4 for the function 1 f(x) = 2x + = x 4
-
3. A tire starts from rest and rolls without slipping downhill at a constant acceleration of 13.6 m/s. The diameter of the tire is 170 cm. (a) Find the angular acceleration of the tire. (b) How long...
-
What is the difference between the DNS local name server and the DNS authoritative name server?
-
What is output of "sh ip bgp sum" command? Explain your answer with an output sample from a network running BGP and analyse the output elements.
-
A manufacturing company produces valves in various sizes and shapes. One particular valve plate is supposed to have a tensile strength of 5 pounds per millimeter (lbs/mm). The company tests a random...
-
The area of a rectangle is 30 cm 2 and its perimeter is 26 cm. Find the length and width of the rectangle.
-
The compound beam is fixed at A, pin connected at B, and supported by a roller at C. Draw the shear and moment diagrams for the beam. 2 kN 3 kN/m |B iA 3 m 3 m
-
Draw the shear and moment diagrams for the compound beam. 5 kN/m I||||..|||| 2 m m
-
An A-36 steel strip has an allowable bending stress of 165 MPa. If it is rolled up, determine the smallest radius r of the spool if the strip has a width of 10 mm and a thickness of 1.5 mm. Also,...
-
How are service-learning projects and CBPR projects alike? How do they differ? Can you think of a situation where one might have advantages over the other? Why is reflection an important part of...
-
Has this client been self-medicating in any way (alcohol, abuse of drugs)? Describe the process that you used to decide about this case. Was this case difficult to find a diagnosis for? Did you...
-
How does climate change occur? What are some of the public health risks of climate change? Which populations are the most vulnerable to climate change health risks? What is the difference between...

Study smarter with the SolutionInn App


