Rank the molecules below in order of increasing boiling point. Cl [T HCH H-C-F 1 -F2 F
Question:
Rank the molecules below in order of increasing boiling point.
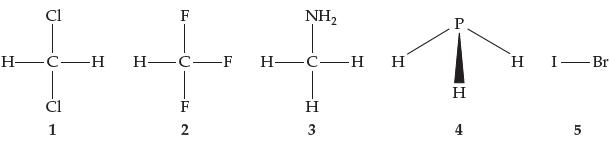
Transcribed Image Text:
Cl [T H—C—H H-C-F 1 -F2 F NH₂ H-CH H H 3 P H 4 H I-Br 5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The molecules in the image can be ranked in order of increasing boiling point as follows 1 ClFNH2 2 ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Rank the following molecules in order of increasing boiling point (without looking up the real values!): (a) 3-methylheptane; (b) Octane; (c) 2,4-dimethylhexane; (d) 2,2,4-trimethylpentane.
-
(a) Rank these molecules in order of increasing boiling point: (b) State your reason for the order you chose in part (a). H H-C-OH H H-H H H-C-H H H-Cl
-
(a) Rank these molecules in order of increasing boiling point: (b) State your reason for the order you chose in part (a). Br Br-C-Br Br Br Br H H-C-H H CI-CI
-
March 31, 2014, adjusted trial balance for Brenner Climbing Adventures has been alphabetized as follows: Required Journalize the closing entries. No. Account Debit Credit $ 2,600 168 Accumulated...
-
You have recently assumed the role of HR Manager in your company. In reviewing the company records, you note that the job descriptions were last updated 5 years ago. The Company President has taken...
-
Select the correct response for each of the following. 1. For the summer session of 20X2, Pacific University assessed its students $1,700,000 (net of refunds) covering tuition and fees for...
-
For the following products and countries, identify the type of warehouse that should be used as well as the method of transportation that should deliver the product to end users. Using the Internet,...
-
Feather Friends, Inc., distributes a high-quality wooden birdhouse that sells for $20 per unit. Variable costs are $8 per unit, and fixed costs total $180,000 per year. Required: Answer the following...
-
Explain the types of anomalies that might occur on this relational model when not normaliz ed NORMALIZATION (40 Points) Consider a COLLEGE logical relational model below....
-
Some bacteria have evolved in such a way to render the antibiotic Vancomycin ineffective. How did they accomplish this (make sure to explain the role of intermolecular forces)?
-
For each part of this problem, redraw both of the molecules, orienting them in a way that allows you to clearly show (using a dotted line) a hydrogen bond between them. If it is not possibleto form a...
-
How important is the global youth market? Suggest products and services they would want to use.
-
Company A buys 100 percent of Company B for $55,000. Accounts Other Assets Investment in Co. B Liabilities Common Stock Retained Earnings Company A $ 90,000 55,000 $145,000 $ 25,000 40,000 80,000...
-
A more recent study of Feline High-Rise Syndrome (FHRS) (see Chapter 1, Example 1.2) included data on the month in which each of 119 cats fell (Vnuk et al. 2004). The data are in the accompanying...
-
Magwitch Limiteds finance director produced the following forecast breakeven chart for the year ending 31 May 2018: During the year the company produced and sold 20,000 units, and both revenue and...
-
The following data are 40 measurements of diameter growth rate of the tropical tree Dipteryx panamensis from a long-term study at La Selva, Costa Rica (Clark and Clark 2012). The data are...
-
The Milky Way appears as a massive band of stars across the night sky. The nocturnal dung beetle (Scarabaeus satyrus) is known to use this band of illumination for orientation. After sculpting a...
-
Obtain from your library a copy of following article: Robert S. Kaplan and David P. Norton, Mastering the Management System, Harvard Business Review (January 2008), pp. 6377. The authors of this...
-
Using Gauss-Jordan elimination, invert this matrix ONLY 0 0 0 0 1
-
Determine the mean free path at 500. K and 1 atm for the following: a. Ne b. Kr c. CH 4 Rather than simply calculating the mean free path for each species separately, instead develop an expression...
-
Consider the diagram of a molecular beam apparatus provided in the text. In the design of the apparatus, it is important to ensure that the molecular beam effusing from the oven does not collide with...
-
A comparison of ave , mp , and rms for the Maxwell speed distribution reveals that these three quantities are not equal. Is the same true for the one-dimensional velocity distributions?
-
Consider a force field (at) F(t) = bt ct and an object of mass m located initially at xo (t = 0)=0=yo ZO The initial velocity is (t = 0) = 0. (a) Compute the kinetic energies at t = 1 and t = 2. (b)...
-
The static budget for Practical Company is based on expected sales of 20,000 units for the upcoming period. At this sales volume, its budgeted income is as follows: Total Sales $1,000,000 Less...
-
To best use Fred's pension, Carlos suggested that his client's Fred and Ethel use pension maximization. This would entail the sale of what type of insurance? Explain.

Study smarter with the SolutionInn App


