What is E forward rxn for the reaction in Problem 13.95? What is E reverse rxn ?
Question:
What is ΔEforward rxn for the reaction in Problem 13.95? What is ΔEreverse rxn?
Data from Problem 13.95
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction?
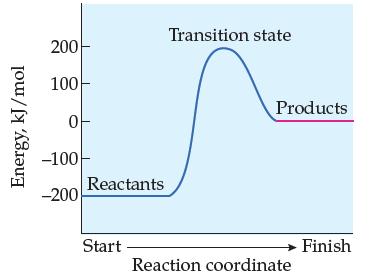
Transcribed Image Text:
Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start Transition state Products Reaction coordinate Finish
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Does the following reaction-energy profile represent an endothermic or exothermic reaction in the forward direction? In the reverse direction? Energy, kJ/mol 200 100- 0 -100 -200 Reactants Start...
-
What is the activation energy for the reverse reaction in Problem 13.95? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in the...
-
What is the activation energy for the forward reaction in Problem 13.95? Data from Problem 13.95 Does the following reaction-energy profile represent an endothermic or exothermic reaction in the...
-
About "Hilton Honors" 1) Explain what creates value and identify if there are aspects that do not create value. (Please include the citation and references) 2)What is the strength of the Hilton Honor...
-
Recording Adjusting and Closing Entries and Preparing a Balance Sheet and Income Statement Including Earnings per Share (AP4-7) Ellis, Inc., a small service company, keeps its records without the...
-
Ebersoll Mining has $6 million in sales, its ROE is 12%, and its total assets turnover is 3.2. The company is 50% equity financed. What is its net income?
-
How many electrons, protons, and neutrons are contained in the following atoms or ions: (a) \({ }^{6} \mathrm{Li}\), (b) \({ }^{13} \mathrm{C}^{+}\), and (c) \({ }^{18} \mathrm{O}^{++}\)?
-
Bliny Corporation makes a product with the following standard costs for direct material and direct labor: During the most recent month, 5,000 units were produced. The costs associated with the months...
-
East Hill Home Healthcare Services was organized on January 1, 2022 by four friends. Each organizer invested $10,000 in the company and, in turn, was issued 8,000 shares. To date, they are the only...
-
From the following reaction-energy profiles, determine whether reactions A and B are exothermic or endothermic in the forward direction: Energy, kJ/mol 300 200 100 0 -100 -200 B Reactants Start...
-
In a kinetic study of the reaction the following rate data were obtained. Write a rate law complete with proper values for the orders. What is the overall order of the reaction? 2 C10(aq)+ 2 OH (aq) ...
-
Andrea is an employee of Fern Corporation. She also has her own business working as a life coach. For 2015, Andrea's wages from Fern were $210,000. Her self-employment income was $30,000. a. Compute...
-
How Goal setting and feedback can be powerful methods for motivating employee?. Give Thorough explanation with references
-
Justify what kind of flow sensor should be used for the cooling water flow through a condenser (approx. 20 L/min at 10C) and what kind of level sensor should be used for a reflux drum ?
-
As a VP of HR for a major supermarket chain, what strategies might be deployed to minimize the risk of violence for employees?
-
Enterprise Rent-A-Car will like to expand its car rental business into the Asian market, particularly Singapore. Analyze the most preferred market entry mode and the economic indices that will make...
-
Identify at least four key principles of successful culture change in the long term care setting.
-
Albert and Patricia are divorced during the current year. As part of their divorce agreement, Patricia agrees to pay Albert alimony of $85,000 in the current year and $5,000 per year in subsequent...
-
Suppose a population of bacteria doubles every hour, but that 1.0 x 106 individuals are removed before reproduction to be converted into valuable biological by-products. Suppose the population begins...
-
Suppose the swimmer in Figure 4.21 has a swimming speed relative to the water of 0.45 m/s and the currents speed is 2.5 m/s. If it takes the swimmer 200 s to cross the river, how wide is the river?...
-
Consider a swimmer who wants to swim directly across a river as in Example 4.7. If the speed of the current is 0.30 m/s and the swimmers speed relative to the water is 0.60 m/s, how long will it take...
-
A swimmer is able to swim at a speed of 0.90 m/s in still water. (a) How long does it take the swimmer to go a distance of 1500 m in still water? Call this time t 1 . (b) The swimmer then decides to...
-
1. Visit the toy section in a large department store such as Target or Walmart. Identify the different sections or aisles of the store (e.g., action figures, dolls and stuffed animals, outdoor play)....
-
what is the price they paid for the bond; if it is a $5000 bond with a coupon % of 2.75 and a yield percentage of 2.62 on a corporate debenture with years of maturity is 9.5
-
6) Suppose that you are allocating your investment capitals between risk free asset and risky asset. The expected returns of risk free and risky assets are 5% and 12%, respectively. The standard...

Study smarter with the SolutionInn App


