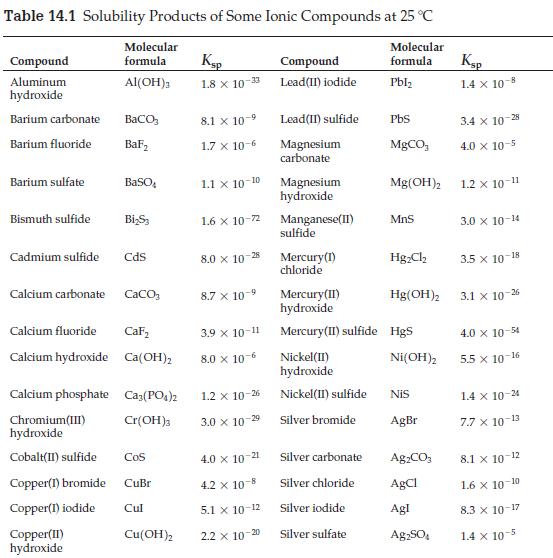
What is the most soluble 1:2 salt in Table 14.1? Explain how you know. Table 14.1 Solubility
Question:
What is the most soluble 1:2 salt in Table 14.1? Explain how you know.

Transcribed Image Text:
Table 14.1 Solubility Products of Some Ionic Compounds at 25 °C Molecular formula Pbl₂ Compound Aluminum hydroxide Barium carbonate Barium fluoride Barium sulfate Bismuth sulfide Cadmium sulfide Calcium carbonate Calcium fluoride Calcium hydroxide Chromium(III) hydroxide Cobalt(II) sulfide Molecular formula Al(OH)3 BaCO3 BaF₂ Copper(II) hydroxide BaSO Bi₂S₂ CdS CaCO3 Calcium phosphate Ca₂(PO4)2 Cr(OH)3 CaF₂ Ca(OH)₂ COS Copper(1) bromide CuBr Copper(1) iodide Cul Cu(OH)2 Ksp 1.8 x 10-33 8.1 x 10-9 1.7 x 10-6 1.1 X 10-10 1.6 x 10-72 8.0 x 10-28 8.7 x 10-⁹ 3.9 X 10-11 8.0 x 10-6 1.2 x 10-26 3.0 x 10-2⁹ 4.0 x 10-21 4.2 x 10-8 5.1 x 10-12 2.2 x 10-20 Compound Lead (II) iodide Lead(II) sulfide Magnesium carbonate Magnesium hydroxide Manganese(II) sulfide Mercury (1) chloride Nickel(II) hydroxide Nickel(II) sulfide Silver bromide PbS MgCO₂ Silver carbonate Silver chloride Silver iodide Silver sulfate Mg(OH)2 MnS Mercury (II) hydroxide Mercury (II) sulfide Hgs Hg₂Cl₂ Hg(OH)2 Ni(OH)2 NIS AgBr Ag,CO, AgCl Agl Ag₂SO4 Ksp 1.4 x 10-8 3.4 x 10-28 4.0 x 10-5 1.2 x 10-11 3.0 x 10-14 3.5 x 10-18 3.1 x 10-26 4.0 x 10-54 5.5 x 10-16 1.4 x 10-24 7.7 x 10-13 8.1 x 10-12 1.6 × 10-10 8.3 x 10-17 1.4 x 10-5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The energy density of gasoline for an ICE car is 43 MJ/kg. The energy density of battery used to power a similar electric car is 3 MJ/kg. Assume that the energy consumption of either the electric or...
-
The table to the right represents the production function for Hawg Wild, a small catering company specializing in barbecued pork. The numbers in the cells represent the number of customers that can...
-
In several of the above exercises, we have indicated that an infinite series 1/(1+r )+1/(1+r )2 +1/(1+r )3 +... sums to 1/r . This can (and has, in some of the B-parts of exercises above) been used...
-
Fletcher, Inc. disposes of under- or over-applied overhead at year-end as an adjustment to cost of goods sold. Prior to disposal, the firm reported cost of goods sold of $619,000 in a year when...
-
Evaluating an Ethical Dilemma Mike Lynch is the manager of an upstate New York regional office for an insurance company. As the regional manager, his compensation package comprises a base salary,...
-
In PC Techs product mix problem, assume there is another PC model, the VXP that the company can produce in addition to Basics and XPs. Each VXP requires eight hours for assembling, three hours for...
-
Determine the number of ideal stages required in Example 7.4 if the solvent rate used is twice the minimum. Data From Example 7.4:- A solution of acetic acid (C) in water (A) is to be extracted using...
-
From historical data, Harry's Car Wash estimates that dirty cars arrive at the rate of 10 per hour all day Saturday. With a crew working the wash line, Harry figures that cars can be cleaned at the...
-
Unusual shapes! At this point, we've gotten pretty comfortable with calculating the perimeter and area of squares and rectangles. In this assignment, we'll make a program that calculates these values...
-
What is dynamic about the equilibrium that is established when a sparingly soluble salt is added to water?
-
Consider a saturated aqueous solution of AgCl, a salt that is only sparingly soluble in water. What happens to this solution if a saturated solution of NaCl (a water-soluble salt) is added to it?
-
Review the advertisements in your local newspaper. Identify products that you perceive to be deals that are too good to be true. These deals are probably examples of potential gray markets (flea...
-
Explain the stages of a successful approach to conflict resolution.?
-
Suppose the House and the Senate pass different versions of a health care reform bill. What kind of committee is responsible for revising each version of the bill into a single bill that both houses...
-
Consider the following code. What is the output? s=0 for a in range (1,5): for b in range (1,4): print(s) s = s + 1
-
Please create a template that includes the variable needed to complete the problem in excel. B) Using the information from your budget (Reference the DATA from your master budget), assume that annual...
-
Explore the concept of allosteric regulation in enzyme kinetics, emphasizing its importance in metabolic pathways and signal transduction cascades
-
Your company has been approached to bid on the contract to sell 17,500 voice recognition (VR) computer keyboards a year for four years. Due to technological improvements, beyond that time they will...
-
AB CORPORATION ISSUED THE FOLLOWING 850 COMMON STOCKS PAR VALUE P100 750 PARTICIPATING PREFERRED STOCKS PAR VALUE P100 AT 3% AB CORPORATION DECLARED P100,000.00 DIVIDEND IN 2022.
-
A passive solar water heater is to be installed on the roof of a multi-story building. The heater tank is open to atmospheric pressure and is mounted 16 m above ground level. In the static...
-
The elevated tank similar to the one shown in Fig. 3.37 is part of a water delivery system to be built for a small village. Find the required elevation of the tank if a minimum gage pressure of 160...
-
In the eye of a hurricane, pressure can sometimes drop from normal atmospheric pressure all the way down to 11 psia. What would be the height reading, in inches, of a mercury barometer there?
-
Identify and describe the 5 most significant product innovation challenges Australian SME's face over the next 5 years. To overcome these challenges Australian SMEs must implement best practices in...
-
Objective: Develop an appropriate promotional mix for Guardians of the galaxy 3. Instructions: Assume the role of promotional/marketing intern for a major motion picture company and you have an...
-
in a paragraph introduce the book Born a Crime, its author and the theme of book or it main idea of the book by explaining the autobiography of comedian Trevor Noah and that in his memoir Noah...

Study smarter with the SolutionInn App


