When a small amount of soap is added to a beaker of water, the soap molecules end
Question:
When a small amount of soap is added to a beaker of water, the soap molecules end up positioning themselves at the surface (hence soaps are often called surfactants):
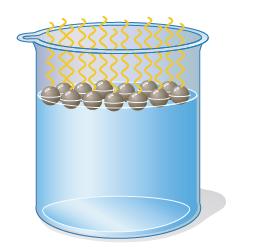
Explain why the soap molecules migrate to the surface, and why they orient themselves with their hydrocarbon tails sticking out of the water.
Transcribed Image Text:
3333
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Soap molecules are amphiphilic meaning they have both hydrophobic waterrepelling and hydrophilic waterattracting parts This amphiphilic nature allows ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
When a pure substance is placed in contact with water, there are three possible outcomes. The substance may do nothing that is, the substance does not dissolve and no visible change takes place. The...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
What approaches to recruitment can an employer adopt in order to create and project a positive public image?
-
Based on the information provided in Problem 11-11A, prepare a schedule of accounts payable for Flints Fantasy as of July 31, 20--. Verify that the accounts payable account balance in the general...
-
Computer upgrade times ( in minutes) are being evaluated. Samples of five observations each have been taken, and the results are as listed. Using factors from Table, determine upper and lower control...
-
Hemlock Semiconductor Operations, LLC, and SolarWorld Industries Sachsen GmbH, are both companies involved in the manufacture of components for solar power products. Prior to the lawsuit, the two...
-
Nelson Company, organized in 2014, has the following transactions related to intangible assets . Instructions Prepare the necessary entries to record these intangibles. All costs incurred were for...
-
What sql commands are used in RDBMS? What are the 5 basic SQL queries?
-
Consider the soap molecules at the top of page 495 and the ionic compound potassium acetate. (a) Draw a dot diagram for the acetate ion, and then place a K + ion next to the O bonded to C by a single...
-
Why do most solids become more soluble in water with increasing temperature?
-
Think of Pasteurs enantiomeric resolution of racemic acid in terms of the resolving agent idea discussed in Sec. 6.8. Did Pasteurs resolution involve a resolving agent? If so, what was it?
-
The market price for a bond that matures in 10 years is $ 900 and its even value is $ 1,000 and pays 8% interest (4% semi-annually). What is the expected rate of return on the bond? Without...
-
Randy purchased a single-family home for $200,000 to use as a rental property. The appraisal of the home valued the building at 76% of the total purchase price. The remaining 24% of the purchase...
-
The multistage counter-current leaching system applies in liquid-liquid extraction as well. Please list THREE other types of liquid-liquid extraction equipment. All different types of extraction...
-
Would evolutionary processes manifest in a population lacking genetic diversity? How would the presence of solely acquired variations influence evolutionary trajectories? Additionally, if all...
-
Assume that, in a Stop-and-Wait ARQ system, the bandwidth of the line is 32 Mbps, and 1 bit takes 16 ms to make a round trip. What is the bandwidth-delay product? If the system data frames are 1500...
-
A study on ethics in the workplace by the Ethics Resource Center and Kronos, Inc., revealed that 35% of employees admit to keeping quiet when they see coworker misconduct. Suppose 75% of employees...
-
Multiple Choice Questions: 1. The largest component of aggregate demand is? a. Government purchases. b. Net exports. c. Consumption. d. Investment. 2. A reduction in personal income taxes, other...
-
If w = 800 lb/ft, determine the absolute maximum shear stress in the beam. The supports at A and B are smooth. 3 ft - 3 ft 6 ft
-
Determine the shear stress at point B on the web of the cantilevered strut at section aa. 4 kN 2 kN 300 mm 250 mm a 250 mm 20 mm 70 mm 20 mm 50 mm
-
Determine the maximum shear stress acting at section aa of the cantilevered strut. 4 kN 2 kN 300 mm 250 mm 250 mm 20 mm 70 mm 20 mm 50 mm
-
The following graph shows the temperature in Nellie's dorm room over a 24 h period. Temperature (C) T 25 20. 15 10- 10 225 5 0 5 10 15 20 25 Time (h) Determine the equation of this sinusoidal...
-
The government supports fast cost recovery through its depreciation policies. The government also supports the contribution of property to a corporation by deferring tax recognition. Are these a fair...
-
a. Determine the prime-power factorizations of both 105 and 189. b. Determine the set of all positive divisors (factors) of 105. c. Determine the set of all positive divisors of 189. d. Determine the...

Study smarter with the SolutionInn App


