Write the equilibrium constant expression for (a) 2 FeCl3(s) + 3 HO(g) (b) FeO3(s) + 3 CO(g)
Question:
Write the equilibrium constant expression for
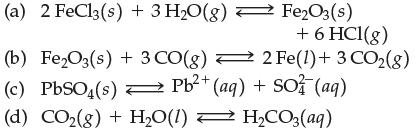
Transcribed Image Text:
(a) 2 FeCl3(s) + 3 H₂O(g) (b) Fe₂O3(s) + 3 CO(g) → (c) PbSO(s) Pb²+ (aq) + SO3(aq) Fe₂O3(s) + 6 HCI(g) 2 Fe(1) + 3 CO₂(g) (d) CO₂(g) + H₂O(1) ⇒ H₂CO3(aq) =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a For the reaction 2FeCl3 s 3H2Og Fe2O3s 6HClg the equilibrium constant expression K is ...View the full answer

Answered By

Qurat Ul Ain
Successful writing is about matching great style with top content. As an experienced freelance writer specialising in article writing and ghostwriting, I can provide you with that perfect combination, adapted to suit your needs.
I have written articles on subjects including history, management, and finance. Much of my work is ghost-writing, so I am used to adapting to someone else's preferred style and tone. I have post-graduate qualifications in history, teaching, and social science, as well as a management diploma, and so am well equipped to research and write in these areas.
4.80+
265+ Reviews
421+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Consider the gas-state reaction (a) Write the equilibrium constant expression for the reaction. (b) Write the reaction in reverse. (c) Write the equilibrium constant expression for the reverse...
-
(a) Write the equilibrium constant expression for the reaction (b) How would the equilibrium be affected if PbI 2 (s) were added? (c) How would the equilibrium be affected if Pb(NO 3 ) 2 (s) were...
-
Compare your equilibrium constant expressions from Problem 14.48(a) and Problem 14.49. (a) Explain how the value of K eq changes for a reaction when you double the reaction (when you multiply it...
-
Tatum is a consultant at R.G. & Company (R.G.), a global consulting firm. She has enjoyed the past few years working at the company. As an ambitious person, she has been focusing on her long-term...
-
Finding Financial Information Refer to the financial statements of American Eagle Outfitters in Appendix B at the end of the book. Required: 1. State the amount of the largest expense on the income...
-
In the pension fund problem, suppose there is a fourth bond, bond 4. Its unit cost in 2010 is $1020; it returns coupons of $70 in years 2011 through 2016 and a payment of $1070 in 2017. Modify the...
-
In the benzene adsorber of Example 9.7, the flow rate is increased to \(0.25 \mathrm{~m}^{3} / \mathrm{s}\). Calculate the breakthrough time and the fraction of the bed adsorption capacity that has...
-
Carlos Arruza Company exchanged equipment used in its manufacturing operations plus $3,000 in cash for similar equipment used in the operations of Tony LoBianco Company. The following information...
-
Given the following data segment: .DATA var1: .BYTE 1, 2, 'Z' stri: .ASCIIZ "My String " var2: .WORD Ox12345678 var3: HALF 1000 Given Varl is 0X10010000 State address of strl, var2 & var3 strl: var2:...
-
As noted in the chapter, the value of K eq for the reaction N 2 (g) + O 2 (g) 2 NO(g) is 0.0017 at 2027 C and 2.3 10 9 at 25 C. (a) Judging from the values of K eq , does this reaction shift to the...
-
What does a catalyst do to the time it takes for a reaction to reach equilibrium? Explain how it does this.
-
Which of the following statements is true? a. Bank robberies are more costly than frauds. b. Fraud is often labeled the fastest growing crime. c. FBI agencies are currently spending approximately 35...
-
What is the power required to accelerate a 10 tons (metric tons) truck from rest to 36 kmh1 in 10 s, on a road level?
-
In Civil Litigation in Kenya what is a Third party proceeding ?
-
The angle between the lockers/wall/elevated surface and the floor is assumed to be a perfect 90 degrees. Explain why?
-
A rock is thrown at a 60.0 angle above the horizontal across level ground. It is thrown from a height of 2.00 m above the ground with a speed of 20.0 m/s and experiences no appreciable air...
-
After completing the process of recording the purchase of supplies, you noticed that two account balances are incorrect in your trial balance related to that transaction. What accounts (by type) are...
-
A company wants to set up a kiosk in the mall and knows that the Fixed Costs will be somewhere between $20,000 and $30,000; they also know that their average selling price will be somewhere between...
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
If the length of the tank in Fig. 4.24 is 1.2 m, calculate the total force on the bottom of the tank. 3 m Tank is Air 200 kPa (gage) 1.2 m long Oil 1.5 m (sg =0.80) Water 2.6 m 2.0 m
-
An observation port in a small submarine is located in a horizontal surface of the sub. The shape of the port is shown in Fig. 4.25. Compute the total force acting on the port when the pressure...
-
A rectangular gate is installed in a vertical wall of a reservoir, as shown in Fig. 4.26. Compute the magnitude of the resultant force on the gate and the location of the center of pressure. Also...
-
How to propose a health care plan and I need to include how I would finance the plan. Elaborate the most better way to manage health care plan discuss all strategies.
-
Discuss the potential disadvantages of brainstorming? How can you overcome the challenges?
-
Explain in your own words the different types of anesthesia, including what types of procedures each are used for.

Study smarter with the SolutionInn App


