If the length of the tank in Fig. 4.24 is 1.2 m, calculate the total force on
Question:
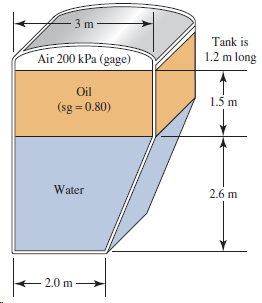
Transcribed Image Text:
3 m Tank is Air 200 kPa (gage) 1.2 m long Oil 1.5 m (sg =0.80) Water 2.6 m 2.0 m
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
F B p B A A 20 12 m 2 24 m 2 p B ...View the full answer

Answered By

Mark Macharia
I am a professional tutor offering academic assistance to students. With a Bachelors Degree in Financial Engineering I majored in complex but common subjects. My majors include:
-Mathematics
-Economics
-Statistics
-Finance
I deliver quality content with zero plagiarism in a timely manner. I have two years experience in the field of academic writing and online technical tutoring.
Some of my technical skills include:
-Microsoft Excel : Statistical analysis & Business Analytics
- R programming : Statistical analysis
- Web Development : HTML 5, CSS3, Bootstrap 4, PHP, Python, and Django.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the total force on the bottom of the closed tank shown in Fig. 4.23 if the air pressure is 52 kPa(gage). Air 0,50 m Oil (sg -0.85) 0.75 m Water 18 m 1.2 m
-
The flat left end of the tank shown in Fig. 4.21 is secured with a bolted flange. If the inside diameter of the tank is 30 in and the internal pressure is raised to +14.4 psig, calculate the total...
-
In Figure a cube of edge length L = 0.600m and mass 450 kg is suspended by a rope in an open tank of liquid of density 1030 kg/m3. Find (a) The magnitude of the total downward force on the top of the...
-
Determine the force P needed to support the 100 lb weight. Each pulley has a weight of 10 lb. Also determine the reactions at A and B. Figure 2 in
-
Tizani SpA's bank statement from Eastern National Bank at August 31, 2017, shows the information below. A summary of the Cash account in the ledger for August shows: balance, August 1, ¬10,559;...
-
A 0.15-g sample of a purified protein is dissolved in water to give 2.0 mL of solution. The osmotic pressure is found to be 18.6 torr at 25 C. Calculate the proteins molar mass.
-
A hypodermic syringe (see Fig. P5.30) is used to apply a vaccine. If the plunger is moved forward at the steady rate of \(20 \mathrm{~mm} / \mathrm{s}\) and if vaccine leaks past the plunger at 0.1...
-
Taxpayer A purchased $ 100,000 of corporate bonds yielding 12.5% per annum; the interest income from these bonds is taxed at a rate of 28%. Taxpayer B purchased $ 100,000 of municipal bonds yielding...
-
Determine the magnitude,direction and position of the force which will keep the body in equilibrium? 20 N 30 30 N 1 m B 1 m 1 m 70 N D 1 m 60 50 N
-
The following diagram describes a service process where customers go through either of two parallel three-step processes and then merge into a single line for two final steps. Capacities of each step...
-
A simple shower for remote locations is designed with a cylindrical tank 500 mm in diameter and 1.800 m high as shown in Fig. 4.22. The water flows through a flapper valve in the bottom through a...
-
Explain the serializable isolation level. Give an example of its use.
-
List the benefits of activity-based costing. How could these reduce costs?
-
Who reported the top 1% householders owned 35.4% of all privately held wealth?
-
Wildhorse Corporation earned $422,000 during a period when it had an average of 100,000 shares of common stock outstanding. The common stock sold at an average market price of $15 per share during...
-
What are important considerations when taking existing machinery and equipment as security?
-
Based on the textbook information included in which you discuss the merits of the cost principle and its importance related to valuation and overall financial statement analysis. In your submission,...
-
As a System Operation Engineer, I have effectively revised the training documents and related supplementary materials for System Operators. This was done to improve the proficiency of ECC system...
-
The Caffeine Coffee Company uses the MIRR. The firm has a cost of capital of 12 percent. The project being analyzed has a $27,000 initial investment and is expected to produce the following case...
-
Chris Zulliger was a chef at the Plaza Restaurant in the Snowbird Ski Resort in Utah. The restaurant is located at the base of a mountain. As a chef for the Plaza, Zulliger was instructed by his...
-
An ideal gas undergoes an expansion from the initial state described by P i , V i , T to a final state described by P f , V f , T in a. A process at the constant external pressure P f . b. In a...
-
Show the reagents you would use to achieve the following transformation:
-
Propose a mechanism for the following transformation: 1) Excess LAH 2) H20 CH3
-
Large-scale industrial production - accompanied by massive technological change, expanding international communication networks, and pro-growth government policies-generated rapid economic...
-
How can this relate to individuals who have had a TBI and are currently homeless or marginally housed?
-
1. According to Marxian theory, are crises an economic process inherent to the accumulation dynamics of current capitalism? Justify your answer. 2. According to Marxian theory, what do you consider...

Study smarter with the SolutionInn App


