Based on the general trends in the periodic table, predict which element in each of the following
Question:
Based on the general trends in the periodic table, predict which element in each of the following pairs has the most metallic character:
(a) Sn or Pb
(b) Ag or Sr
(c) Al or B
(d) Ga or Ge.
Periodic Table:
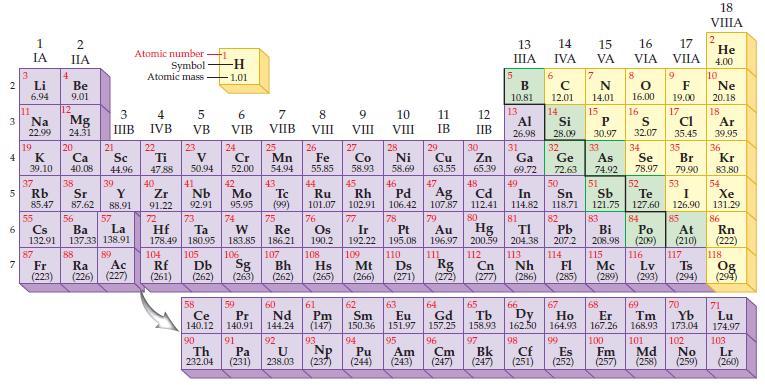
Transcribed Image Text:
2 3 4 5 10 6 7 3 11 Li 6.94 1 IA Na 22.99 19 K 39.10 37 87 4 Fr (223) 2 IIA Be 9.01 12 Mg 24.31 20 Ca 40.08 38 21 Rb Sr Y 85.47 87.62 88.91 88 3 IIIB Sc 44.96 39 55 56 La Cs Ba 132.91 137.33 138.91 57 89 Ra Ac (226) (227) Atomic number Symbol Atomic mass 4 IVB 22 Ti 47.88 40 Zr 2 91.22 72 5 VB 104 23 V 50.94 41 73 Hf Ta 178.49 180.95 105 Rf Db (261) (262) -H -1.01 6 VIB Nb Mo 92.91 95.95 90 24 Cr 52.00 42 74 106 Sg (263) 58 Pr Ce 140.12 140.91 91 59 7 VIIB Th Pa 232.04 (231) 25 W Re 183.85 186.21 Mn 54.94 43. Tc (99) 75 107 Bh (262) 60 Nd 144.24 92 U 238.03 8 VIII 26 Fe 55.85 76 Os 190.2 61 Pm (147) 93 9 VIII Np (237) 10 VIII 11 IB 12 IIB 27 28 29 Zn Co Ni Cu 58.93 58.69 63.55 65.39 30 5 44 47 48 49 45 46 Ru Rh Pd Ag Cd In 101.07 102.91 106.42 107.87 112.41 114.82 77 78 79 80 Ir Pt 192.22 195.08 109 110 Hs Mt Ds Rg Cn (265) (266) (271) (272) (277) Au Hg 196.97 200.59 111 112 108 13 ΠΙΑ B 10.81 13 Ga 69.72 6 14 IVA с 12.01 Al Si 26.98 28.09 32 31 Ge 72.63 14 81 82 TI Pb 204.38 207.2 113 114 Nh Fl (286) (285) 15 16 17 VA VIA VILA 66 67 62 63 64 65 Sm Eu Gd Tb Dy Ho 150.36 151.97 157.25 158.93 162.50 164.93 94 95 96 97 Pu Am Cm Bk (244) (243) (247) (247) 7 N 14.01 15 P 30.97 8 68 O 16.00 33 34 50 As Se 74.92 78.97 51 52 Sn Sb Te I 118.71 121.75 127.60 126.90 53 83 84 85 Bi Po At 208.98 (209) (210) 115 116 117 Lv Ts (289) (293) (294) Mc 98 99 167.26 100 Cf Es Fm (251) (252) (257) 16 S 32.07 9 F 19.00 17 Cl 35.45 35 69 70 Er Tm Yb 168.93 173.04 101 102 Md No (258) (259) Br 79.90 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39,95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
a P...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Based on the general trends in the periodic table, predict which element in each of the following pairs has the smaller atomic radius: (a) Na or K (b) P or N (c) Ca or Ni (d) Si or S. Periodic Table:...
-
Based on the general trends in the periodic table, predict which element in each of the following pairs has the higher ionization energy: (a) Li or Na (b) O or F. Periodic Table: 2 3 4 in 6 3 7 Li...
-
Based on the general trends in the periodic table, predict which element in each of the following pairs has the higher ionization energy: (a) Na or Mg (b) O or S. Periodic Table: 2 3 4 15 6 7 3 11...
-
What is the difference between a Type I error and a Type II error?
-
Performance Measures. Information from the Form 990 for the American Heart Association for the fiscal year ending June 30, 2007, follows. Required a. Compute the following performance measures using...
-
What is an exchange rate? What is the difference between direct and indirect rates? What is a cross rate?
-
Light with a wavelength of \(375 \mathrm{~nm}\) illuminates a metal cathode. The maximum kinetic energy of the emitted electrons is \(0.76 \mathrm{eV}\). What is the longest wavelength of light that...
-
Bernies Bike Shop receives the following trade discounts: 35/25/15. The vendors price list indicates that 35 percent off list price is for purchasing bikes in quantities of 100 or more, 25 percent...
-
Compute the student's GPA for the term. Round your answer to the nearest hundredth. A = 4 points, B = 3 points, C = 2 points, D = 1 point, F = 0 points Course Credits Grade MAT082 3 B ENG071 4 A...
-
What is the group number for each of the following families of elements? (a) Alkali metals (b) Alkaline earth metals (c) Halogens (d) Noble gases.
-
Why did Mendeleev not include argon in his periodic table of 1871? Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 Cs 132.91 87 4 Fr (223) 2 IIA Be 9.01 12 Mg 24.31...
-
The common stock of Alexander Hamilton Inc. is currently selling at $120 per share. The directors wish to reduce the share price and increase share volume prior to a new issue. The per share par...
-
Identify and briefly describe the four elements of a firms marketing mix.
-
Determine specifically what the requirements are for starting a limited liability company in your state. Indicate what forms need to be filed, where they can be obtained, how the filing process...
-
Joshua Sherman, who lives near Vancouver, Canada, is preparing to pitch an idea for a social gaming Web site to a group of angel investors. The site has some new and novel aspects to it. He made a...
-
What are the six steps in applying for a patent? Make your answer as thorough as possible.
-
What are the personal attributes that affect a founders chances of launching a successful new firm? In your judgment, which of these attributes are the most important? Why?
-
Determine the equation of the least squares regression line to predict y from the following data. a. Construct a 95% confidence interval to estimate the mean y value for x = 60. b. Construct a 95%...
-
A horizontal annulus with inside and outside diameters of 8 and 10 cm, respectively, contains liquid water. The inside and outside surfaces are maintained at 40 and 20oC, respectively. Calculate the...
-
Where can authoritative iGAAP guidance be found related to cash and receivables?
-
Briefly describe some of the similarities and differences between U.S. GAAP and iGAAP with respect to the accounting for cash and receivables.
-
Simonies Company, which uses iGAAP, has a note receivable with a carrying value of $30,000 at December 31, 2010. On January 2, 2011, the borrower declares bankruptcy, and Simonis estimates that only...
-
solve with explanation please. 3 (15 points) Apply the list-processing algorithm with two machines using the priority list 77.TTTT. T, Ts and the ordered-requirement digraph to determine the time it...
-
Perform the first three steps that you would do to sort the array using mergesort. When splitting up the array or a subarray, if the (sub)array size is odd, make the left half of the subarray the...
-
*Javascript Requirements: You are not creating a visualization of Conway's Game of Life. You are simply writing a function named stepBoard that takes a 2D array of booleans and returns a 2D array of...

Study smarter with the SolutionInn App


