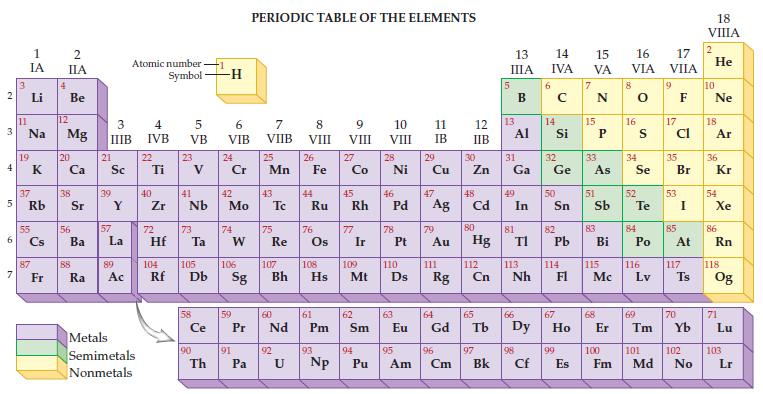
Given the only naturally occurring isotope of fluorine is F-19, determine its mass from the periodic table.
Question:
Given the only naturally occurring isotope of fluorine is F-19, determine its mass from the periodic table.
Transcribed Image Text:
2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -1 Th -H 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 Re 60 P Pr Nd 92 8 VIII U 26 44 Fe Ru 76 61 Pm 93 9 VIII 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Np 27 Co 45 Rh 77 Ir 62 Sm 94 10 VIII Pu 28 Ni 46 78 47 Pd Ag 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 31 Ga Al 49 81 In 66 TI 113 Nh 98 Cf 6 14 15 16 IVA VA VIA 14 Si 32 Ge 50 82 Pb 114 67 99 E Es 15 33 As 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 Cl 35 53 85 Br I At 117 70 Ts 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Fluorine 19F Solution ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Given the only naturally occurring isotope of phosphorus is P-31, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB...
-
Given the only naturally occurring isotope of aluminum is Al-27, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
Given the only naturally occurring isotope of sodium is Na-23, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
In Exercises 1138, use the given conditions to write an equation for each line in point-slope form and slope-intercept form. Slope = -5, passing through (-4, -2)
-
Define fixed cost and variable cost and give an example of each.
-
In June 2007, General Electric (GE) had a book value of equity of $117 billion, 10.3 billion shares outstanding, and a market price of $38.00 per share. GE also had cash of $16 billion, and total...
-
A sports reporter thinks that the median weight of offensive linemen in the NFL is greater than 300 pounds. He obtains a random sample of 30 offensive linemen and obtains the data shown in Table 4....
-
Why does Harrahs system work so well compared to MIS efforts by other companies? Joseph, a 30-something New Yorker, recently went on a weekend trip to Atlantic City, New Jersey, where he hoped to...
-
Equivalent Capacitance - 4 5 Calculate the equivalent capacitance between points a and b in the combination of capacitors shown in the figure below (in F). Use C = 3.92 F, C = 7.16 F, C3 = 5.15 F and...
-
Calculate the atomic mass for lithium given the following data for its natural isotopes. 'Li: 7Li: 6.015 amu 7.016 amu 7.42% 92.58%
-
Draw a diagram of the arrangement of protons, neutrons, and electrons in an atom of each of the following isotopes. (a) 31 15 P (b) 35 17 Cl (c) 40 18 Ar (d) 131 5 3 I.
-
On June 30, the end of the current fiscal year, the following information is available to Axel Companys accountants for making adjusting entries: a. Among the liabilities of the company is a mortgage...
-
One of the software products that supports Critical Chain is based on the Oracle database. Describe one major advantage of having a project management product use the Oracle database and one major...
-
What obligations does the auditor have regarding communicating strengths or weaknesses in internal controls?
-
What are the three elements of an operational audit?
-
How should a resource manager decide which task a resource should work on, when there is a conflict?
-
What are the three most common theories used to explain the origins of the demand for audit and assurance services?
-
A, a middle-level manager of Drug Company X, has been told by her boss, B, to change some figures on the percentage of rats that died as a result of injections of a new drug for which Company X is...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
What is the present value cost of owning the equipment? set up a time line which shows the net cash flows over the period t = 0 to t = 4, and then find the PV of these net cash flows, or the pv cost...
-
Explain the rationale for the discount rate you used to find the PV. MINI CASE Lewis Securities Inc. has decided to acquire a new market data and quotation system for its Richmond home office. The...
-
What is Lewiss present value cost of leasing the equipment? MINI CASE Lewis Securities Inc. has decided to acquire a new market data and quotation system for its Richmond home office. The system...
-
To understand the underlying dynamics, use a relevant strategic framework, such as the 3Cs or Porter's Five Forces, to inspect the problem and analyse the situation. 2. Estimate the DK's gross profit...
-
Here is the demographic pyramid for South Korea. The pyramid illustrates the size of the different age cohorts in a country. The typical shape is, precisely, that of a pyramid. As you can see, in the...
-
As is almost always the case in Macro, there are at least two camps discussing whether the Fed ought to initiate a process of rate cuts sometime in the first half of 2024 and those that think it...

Study smarter with the SolutionInn App


