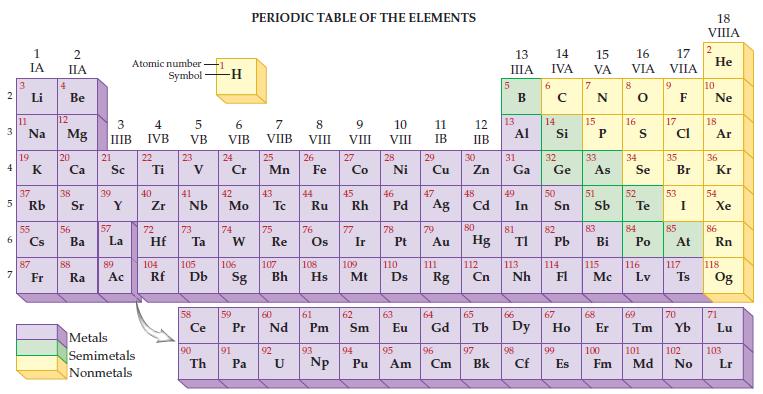
Given the only naturally occurring isotope of phosphorus is P-31, determine its mass from the periodic table.
Question:
Given the only naturally occurring isotope of phosphorus is P-31, determine its mass from the periodic table.
Transcribed Image Text:
2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -1 Th -H 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 Re 60 P Pr Nd 92 8 VIII U 26 44 Fe Ru 76 61 Pm 93 9 VIII 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Np 27 Co 45 Rh 77 Ir 62 Sm 94 10 VIII Pu 28 Ni 46 78 47 Pd Ag 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 31 Ga Al 49 81 In 66 TI 113 Nh 98 Cf 6 14 15 16 IVA VA VIA 14 Si 32 Ge 50 82 Pb 114 67 99 E Es 15 33 As 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 Cl 35 53 85 Br I At 117 70 Ts 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The mass of an element listed on the periodic table ...View the full answer

Answered By

Emel Khan
I have the ability to effectively communicate and demonstrate concepts to students. Through my practical application of the subject required, I am able to provide real-world examples and clarify complex ideas. This helps students to better understand and retain the information, leading to improved performance and confidence in their abilities. Additionally, my hands-on approach allows for interactive lessons and personalized instruction, catering to the individual needs and learning styles of each student.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Given the only naturally occurring isotope of fluorine is F-19, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
Given the only naturally occurring isotope of aluminum is Al-27, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
Given the only naturally occurring isotope of sodium is Na-23, determine its mass from the periodic table. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21...
-
In Exercises 1126, determine whether each equation defines y as a function of x. x + y = 16
-
How can knowing cost behavior relative to volume fluctuations affect decision making?
-
Chen Brothers, Inc., sold 4 million shares in its IPO, at a price of $18.50 per share. Management negotiated a fee (the underwriting spread) of 7% on this transaction. What was the dollar cost of...
-
Use Minitab to obtain the results of Tukey's test for the shear bond strength data from Example 1 in Section 13.1. Approach The steps for obtaining the results of Tukey's test using Minitab, Excel,...
-
A partially completed worksheet for At Home Pet Grooming Service, a firm that grooms pets at the owner's home, follows. INSTRUCTIONS 1. Record balances as of December 31 in the ledger accounts. 2....
-
Calculate the mass flow rate of R-134a through the low-pressure and high-pressure evaporators, each in kg/s. (b) Determine the mass flow rate, in kg/s, specific enthalpy, in kJ/kg, and temperature,...
-
Calculate the atomic mass for lithium given the following data for its natural isotopes. 'Li: 7Li: 6.015 amu 7.016 amu 7.42% 92.58%
-
Draw a diagram of the arrangement of protons, neutrons, and electrons in an atom of each of the following isotopes. (a) 31 15 P (b) 35 17 Cl (c) 40 18 Ar (d) 131 5 3 I.
-
True or False: One limitation of the for loop is that only one variable may be initialized in the initialization expression.
-
Why is it preferred that most communications with those charged with governance be done in writing?
-
Should the length of a program have an impact on whether to set up a separate project or task for administrative support? How about for raw materials?
-
Describe the five types of buffers used by the Critical Chain methodology, and the unique purpose of each one.
-
Describe the primary role of the drum manager.
-
What is the objective of a financial statement audit?
-
B knows that 200 percent cost overruns exist because of the negligence of Company Y's management in carrying out a federal government contract to build an airplane. B works in the comptroller's...
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
How are leases classified for tax purposes? MINI CASE Lewis Securities Inc. has decided to acquire a new market data and quotation system for its Richmond home office. The system receives current...
-
What effect does leasing have on a firms balance sheet? MINI CASE Lewis Securities Inc. has decided to acquire a new market data and quotation system for its Richmond home office. The system receives...
-
What effect does leasing have on a firms capital structure? MINI CASE Lewis Securities Inc. has decided to acquire a new market data and quotation system for its Richmond home office. The system...
-
1.Why do neoliberals argue in support of an unregulated market, and why do they think of the subject in terms of homo economics? What do you see as the central problem with the neoliberal conception...
-
What is inflation? Was it a problem during colonial times? When does inflation negatively impact income and wealth? When does it ease problems of debtors against creditors? When does it encourage...
-
If demand for built space in real estate is declining, does this imply that real rents (that is, rents measured net of inflation) will fall in the market? Why or why not? As in the previous question,...

Study smarter with the SolutionInn App


