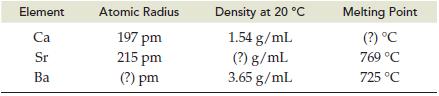
Predict the missing value (?) for each physical property listed below. The (a) Atomic radius, (b) Density,
Question:
Predict the missing value (?) for each physical property listed below. The
(a) Atomic radius,
(b) Density,
(c) Melting point are given for two of three alkaline earth metals in Group IIA/2.
Transcribed Image Text:
Element Ca Sr Ba Atomic Radius 197 pm 215 pm (?) pm Density at 20 °C 1.54 g/mL (?) g/mL 3.65 g/mL Melting Point (?) °C 769 °C 725 °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
We can estimate a value for the physical property of an element by observing the trend in values for ...View the full answer

Answered By

Simranjeet kaur Sangatni
Accountancy is my favorite subject. I worked as Asst. Accountant in Sanjay Pandya Accountancy Firm and also as Faculty of Accountancy in Reliable Eduworld for 11th and 12th CBSE and for CA CPT entrance. I used to take private classes also for 12th and B.com students. I also worked as freelancing in solution providing company.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
What are the expected readings of the ammeter and voltmeter for the circuit inFigure? 6.00 2 10.0 2 6.00 V 5.00 2 4.50 V 6.00 2
-
Predict the missing value (?) for each physical property listed below. The (a) Atomic radius, (b) Density, (c) Melting point are given for two of the metals in Group VIII/10. Element Ni Pd Pt Atomic...
-
Predict the missing value (?) for each property listed below. The atomic radius, density, and melting point are given for elements in Group VIII/8. Element Fe Ru Os Atomic Radius 126 pm (?) pm 135 pm...
-
When Ralph Lauren makes shirts to a customers exact preferences, what utility is provided?
-
What is an annuity agreement and how does it differ from a life income fund?
-
What are some advantages of matching the maturities of claims against assets with the lives of the assets financed by those claims? Is it feasible for a firm to match perfectly the maturities of all...
-
A neutron is shot straight up with an initial speed of \(100 \mathrm{~m} / \mathrm{s}\). As it rises, does its de Broglie wavelength increase, decrease, or not change? Explain.
-
Consider the following independent situations. Situation 1: Ducharme Corporation purchased electrical equipment at a cost of $12,400 on June 2, 2011. From 2011 through 2014, the equipment was...
-
22. Cesar wants to draw a graph representing how he spends each hour of the day. He says that he can't use a circle graph because there are 24 hours in a day, not 100. How should you respond?
-
Why did Mendeleev not include einsteinium in his periodic table of 1871? Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 Sr...
-
Given the density of silver (10.5 g/cm 3 ) and gold (19.3 g/cm 3 ) in Group IB/11, estimate the density for the synthetic element roentgenium, which is below gold in the periodic table. Periodic...
-
James Walker operates a store that sells computer software. Walker has agreed to enter into a partnership with Robert Tolliver, effective January 1, 20X1. The new firm will be called International...
-
Define the term core competency and describe why its important for a firm to have one or more core competencies. How do a companys core competencies help shape its business model?
-
Amanda King has spent the past 15 years as a consultant for a national consulting company. Shes worked with a range of organizations, from Fortune 500 companies to start-ups to nonprofits. Recently,...
-
Describe the most common sources of equity funding.
-
Select a company that has a Twitter account established specifically for the purpose of recruiting. Follow the site for a week or look at its archive of postings and read the tweets for the past...
-
Zappos.com is an online shoe and apparel retailer that has built a strong brand and has shown impressive sales growth since its founding. Its based in Henderson, Nevada, just outside Las Vegas. The...
-
People in the aerospace industry believe the cost of a space project is a function of the weight of the major object being sent into space. Use the following data to develop a regression model to...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Teresa Ramirez and Lenny Traylor are examining the following statement of cash flows for Pacific Clothing Stores first year of operations. Teresa claims that Pacifics statement of cash flows is an...
-
Ashley Company is a young and growing producer of electronic measuring instruments and technical equipment. You have been retained by Ashley to advise it in the preparation of a statement of cash...
-
Each of the following items must be considered in preparing a statement of cash flows for Cruz Fashions Inc. for the year ended December 31, 2010. 1. Fixed assets that had cost $20,000 612 years...
-
A member firm is required to verify the identification of ti customers and run a check in accordance with regulations established by OFAC . What is OFAC and the purpose of the I.D. check?
-
ASAP PLEASE: Electoral Process Management System (EPMS) We want to design a data management application on the electoral process. We assumed that the data covers only one election. All eligible...
-
Using REST technology / IDE is open. You are allowedto use any platform of your choice. Using an IDE of your choice, create a Java project and configure it to be a REST webservice provider. Add an...

Study smarter with the SolutionInn App


