Refer to Figure 12.9 and indicate which of the following are nonpolar covalent bonds. (a) ClCl (b)
Question:
Refer to Figure 12.9 and indicate which of the following are nonpolar covalent bonds.
(a) Cl—Cl
(b) Cl—N
(c) N—H
(d) H—P.
Figure 12.9
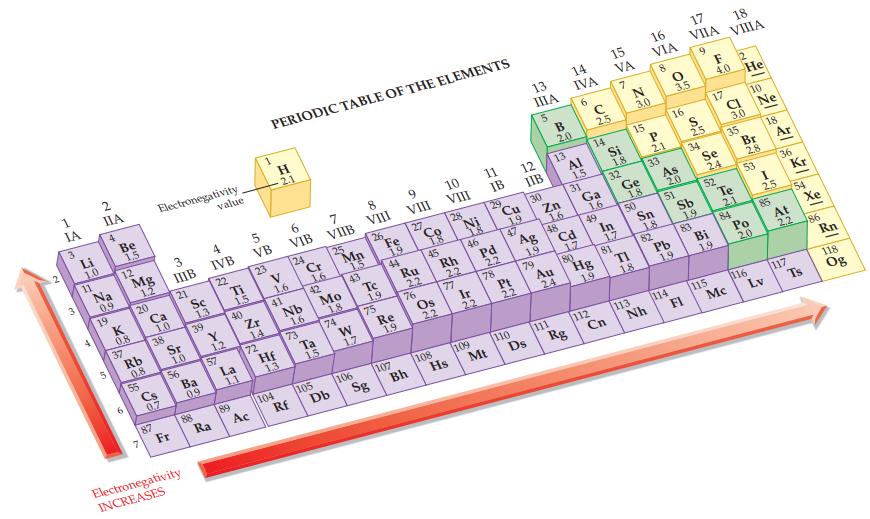
Transcribed Image Text:
IA 3 3 Li 1.0 11 4 IIA Bo Na 0.9 19 5 Be 1,5 12 NE K 0.8 37 Mg 1.2 20 Rb 0.8 55 Ca 1.0 Electronegativity 38 Cs 0,7 87 3 IIIB 21 Sr 1.0 56 Fr Sc 1.3 39 Ba 0.9 Electronegativity INCREASES 88 value 4 IVB 22 Y 1.2 57 Ra Ti 15 89 40 La 1.1 5 VB 23 Zr 14 72 Ac PERIODIC TABLE OF THE ELEMENTS H 2.1 V 1.6 41 Hf 13 104 6 VIB 24 Nb 16 73 Rf Cr 1.6 42 Ta 1.5 7 VIIB 105 25 Mo 1.8 74 Db Mn 1.5 43 W 1,7 8 VIII 106 26 Te 19 75 Sg Fe 22 EN ON 19 44 Re 1.9 9 VIII 107 27 Ru 22 76 Bh Co 45 Os 22 10 VIII 28 108 Rh 22 77 Hs 18 46 Ir 22 11 IB 109 29 Pd 2.2 78 Mt Cu 19 47 Pt 22 110 13 ΠΙΑ 5 12 IIB 30 Ag 1.9 79 Ds B 2.0 Zn 13 16 48 Au 111 24 14 IVA 6 Al 1.5 31 Cd 17 Rg 80 с 2.5 14 Ga 1.6 49 112 Hg 19 15 VA 7 Si 1.8 Cn 32 In 1.7 81 Ge 21 1.8. 15 50 7 TI 18. 3.0 113 16 VIA Nh P 2.1 Sn 18 8 33 82 As 20 Pb 1.9 O 3.5 51 114 17 VIIA 9 16 S 25 34 Sb 1.9 FI 83 Se 24 Bi F He 4.0 17 52 115 19 18 VIIIA CI 3.0 35 Te 2.1 Mc 84 Br 10 2.8 53 Po 20 116 Ne 18 I 25 85 3 Lv Ar 36 At Kr 117 54 2.2 Xe Ts 86 Rn 118 Og
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The correct answer a ClCl Reason a ClCl Chlorine Cl has the same electronegati...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to Figure 12.9 and indicate which of the following are nonpolar covalent bonds. (a) IC (b) CS (c) SH (d) HBr. Figure 12.9 IA 3 3 Li 1.0 11 4 IIA Bo Na 0.9 19 5 Be 1,5 12 NE K 0.8 37 Mg 1.2 20...
-
Refer to the electronegativity values in Figure 12.9 and predict which of the following bonds is least polar: CN, CO, or CI. Figure 12.9 1 IA 2 3 3 Li 1.0 11 4 2 IIA 4 Na 0.9 19 10 5 Be 1,5 12 K 0.8...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Please answer the following. Show your calculations for potential partial credit. Assume the expected return on the market is 14 percent and the risk-free rate is 4 percent. A.)What is the expected...
-
1. Depreciation expense accounts would likely be found in the a. General fund b. Capital projects fund c. Debt service fund d. Enterprise fund 2. The government-wide statements of a state government...
-
The Gulfstream 100 is an executive jet that seats six, and it has a doorway height of 51.6 in. a. What percentage of adult men can fit through the door without bending? b. Does the door design with a...
-
The top left portion of Figure P27.35 shows a currentcarrying wire shaped into a rectangular loop that is very flexible. The loop is mounted on a base (not shown) that allows it to spin in any...
-
Clorox Company is a leading producer of laundry additives, including Clorox liquid bleach. In the 6 months ended December 31, 2011, net sales of $2,526 million produced net earnings of $235 million....
-
19 20 Assertion A compass needle is placed near a current carrying wire. The deflection of the compass needle decreases when the magnitude of the current in the wire is increased. Reason The strength...
-
Which of the following elements occur naturally as diatomic molecules: H, N, Cl, Br, Ar?
-
Refer to Figure 12.9 and label each atom in the following polar covalent bonds using delta notation ( + and ). (a) HS (b) OS (c) NF (d) SCl. Figure 12.9 IA 3 3 Li 1.0 11 4 IIA Bo Na 0.9 19 5 Be 1,5...
-
In Problems 4152, the graph of an equation is given. (a) Find the intercepts. (b) Indicate whether the graph is symmetric with respect to the x-axis, the y-axis, the origin or none of these. -3 3 -3...
-
A factory equipment is purchased on January 6 , 2 0 2 4 , for $ 2 0 , 0 0 0 cash and a $ 5 0 , 0 0 0 note payable. Related cash expenditures include insurance during shipping, $ 5 0 0 ; the annual...
-
Frito-Lay sells a wide range of products, such as Lay's and Ruffles potato chips, Doritos tortilla chips, Tostitos tortilla chips and dips, and Cheetos. Its audience includes families with children...
-
Explain working of K nearest algorithm.
-
The three lines on the graph are the same gas (both type and amount) for different temperatures. I will refer to them as either solid, (-100), dashed (20) and dotted (600). The Y-axis is the number...
-
Suppose that an investor buys a 180-day Australian bank bill with quoted yield 4.2% and sells the bill after 30 days when the quoted yield is 4.2% (on a 150-day bill). What is the holding period...
-
Pauline's Pastry Shop decides to remodel its offices this year. As part of the remodeling, Pauline's trades furniture with a cost of $12,000 that had been expensed in the year of purchase (Section...
-
Outline a general process applicable to most control situations. Using this, explain how you would develop a system to control home delivery staff at a local pizza shop.
-
Costs of borrowing youve worked out a line of credit arrangement that allows you to borrow up to $50 million at any time. The interest rate is .72 percent per month. In addition, 4 percent of the...
-
Costs of Borrowing A bank offers your firm a revolving credit arrangement for up to $80 million at an interest rate of 1.90 percent per quarter. The bank also requires you to maintain a compensating...
-
Cash Management Policy rework Problem 15 assuming: a. Wildcat maintains a minimum cash balance of $50 million. b. Wildcat maintains a minimum cash balance of $20 million. Based on you answer in (a)...
-
presentation - You will author a 10 - 12 slide PowerPoint presentation on the following topic "A Personal Account of My Engagement with Operations Management ." Eh. You are expected to draw upon your...
-
Suppose the Bank of England's repo rate is 4.5% and the financial pages in the newspaper contain the following information: [Tripos 2006] UK GILTS Interest Redemption yield yield Tr 72pc '06 7.32...
-
I. Define Public Relations in this Context: In the given scenario, what does the term "Public Relations" mean to you? How would you articulate the role of PR in managing the fallout of a...

Study smarter with the SolutionInn App


