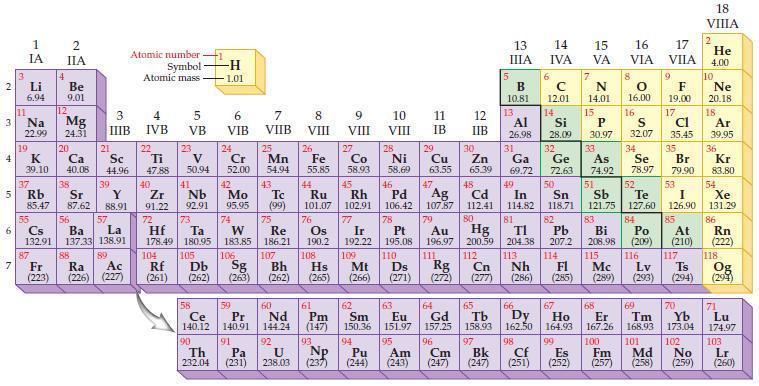
Refer to the periodic table and select the symbol of the element that fits each of the
Question:
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions.
(a) The semimetal in the third period
(b) The semimetal in the fourth period and Group VA/15
(c) The halogen that exists as a reddish-brown liquid
(d) The noble gas that is radioactive.
Periodic Table:
Transcribed Image Text:
2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 Sr 85.47 87.62 20 38 Ca Sc 40.08 44.96 21 56 Cs La Ba 132.91 137.33 138.91 88 3 IIIB 39 Y 88.91 57 89 Atomic number Symbol Atomic mass Ra Ac (226) (227) 4 IVB 22 Ti 47.88 40 Zr 91.22 72 5 VB 23 V 50.94 41 Nb 92.91 73 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 104 105 Rf Db (261) (262) (263) Sg 106 59 Pr 140.91 91 7 VIIB Hf Ta W Re 178.49 180.95 183.85 186.21 Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 Fe Co 55.85 58.93 Os 190.2 44 45 Ru Rh 101.07 102.91 76 9 VIII 61 Pm (147) 27 108 Bh Mt Hs (262) (265) (266) 93 U NP 238.03 (237) 77 Ir 192.22 109 10 VIII 62 Sm 150.36 94 28 Ni 58.69 46 Pd 106.42 78 11 IB 12 IIB 13 IIIA 5 B 10.81 13 Al 26.98 14 15 IVA 6 C 12.01 14 Si 28.09 32 17 16 VA VIA VIIA 8 7 N 14.01 15 P 30.97 79 Pt 195.08 110 Ds 81 82 Au Hg TI Pb 196.97 200.59 204.38 207.2 111 112 113 114 Rg Cn Nh Fl (271) (272) (277) (286) (285) 29 30 31 33 As Ge Se Cu Zn Ga 63.55 65.39 69.72 72.63 74.92 78.97 16.00 16 S 32.07 34 47 51 48 49 50 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 83 Bi 208.98 115 84 Po (209) 9 F 19.00 Md (258) 17 Cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 117 116 Mc Ts Lv (289) (293) (294) 66 67 70 63 64 65 68 69 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 98 99 100 Cf Es Fm (251) (252) (257) 95 101 102 Pu Am (244) (243) Cm (247) 97 Bk (247) 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a The semimetal in the third period is Si sili...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The semimetal in the second period (b) The semimetal in the fourth period and Group...
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The fourthperiod alkali metal (b) The fourthperiod alkaline earth metal (c) The rare...
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The thirdperiod alkali metal (b) The thirdperiod alkaline earth metal (c) The rare...
-
Certain companies regard the management of cultural diversity as a way of developing new competencies that give them certain advantages. They are, for example, able to recruit and hold on to good...
-
Internet Case. The City of Belleview receives pass-through funds from the states Department of Housing to assist in administering the federally funded Supportive Housing Program for the elderly. At...
-
The company had the following shares of stock outstanding during the year. Data regarding dividend privileges and net income are also given. Common shares outstanding: 320,000 for the entire year...
-
RQMs return on investment is: (a) 22.5%. (b) 26.5%. (c) 19.5%. (d) 40.5%. RQM Corporation Selected Financial Data, December 31, 2024 Net sales $1,800,000 Cost of goods sold 1,080,000 Operating...
-
Marriott International is a worldwide operator and franchisor of hotels and related lodging facilities totaling over $1.4 billion in property and equipment. It also develops, operates, and markets...
-
For problems 5 and 6, find the sum of the series described. 5. a, 2, r=5, ^=10 6. 2+ 6+ (-18) + n=12
-
According to IUPAC convention, what is the designation for each of the following groups of elements? (a) Group IA (b) Group IIIA (c) Group VA (d) Group VIIA (e) Group IB (f) Group IIIB (g) Group VB...
-
Identify the group number corresponding to each of the following families of elements. (a) Boron group (b) Oxygen group (c) Nickel group (d) Copper group.
-
JDM Inc. made the following prepayments for expense items during 2019: a. Prepaid building rent for 1 year on April 1 by paying $6,600. Prepaid rent was debited for the amount paid. b. Prepaid 12...
-
How do menu costs contribute to sticky prices?
-
As an input to production, how does technology differ from labor and capital inputs?
-
On April 1, Jiro Nozomi created a new travel agency, Adventure Travel. The following transactions occurred during the companys first month. Required 1. Use the balance column format to set up each...
-
Distinguish between a closed and an open economy. How do the conditions required for goods market equilibrium differ in the two types of economies?
-
One of the most well-known population control policies is the one-child policy implemented by China since the late 1970s. Comment on the side effects of such a policy. This policy has been dubbed a...
-
The U.S. Bureau of Labor Statistics produces consumer price indexes for several different categories. Shown here are the percentage changes in consumer price indexes over a period of 20 years for...
-
What mass of H2 will be produced when 122 g of Zn are reacted? Zn(s) + 2HCl(aq) ( ZnCl2(aq) + H2(g)
-
At December 31, 2010, Besler Corporation had a projected benefit obligation of $560,000, plan assets of $322,000, and prior service cost of $127,000 in accumulated other comprehensive income....
-
Shin Corporation had a projected benefit obligation of $3,100,000 and plan assets of $3,300,000 at January 1, 2010. Shin also had a net actuarial loss of $465,000 in accumulated OCI at January 1,...
-
Hawkins Corporation has the following balances at December 31, 2010. Projected benefit obligation $2,600,000 Plan assets at fair value 2,000,000 Accumulated OCI (PSC) 1,100,000 How should these...
-
The following table lists balance of payment current accounts for Country A. Current Accounts 1. Exports of goods, services, and income 2. Goods 3. Services 4. Income receipts on U.S. assets abroad...
-
What is common law? What is the advantage of common law and why is it an important aspect of the legal system?
-
he shares of firms with diversified operations are ________. Question 7 options: generally positively affected by diversification, because of the reduction in risk generally positively affected by...

Study smarter with the SolutionInn App


