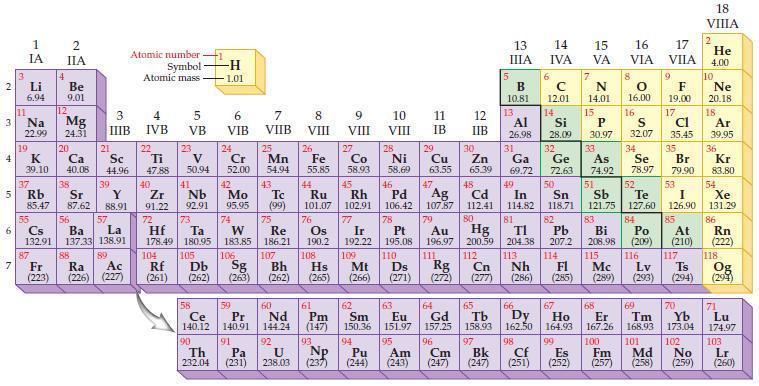
Refer to the periodic table and select the symbol of the element that fits each of the
Question:
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions.
(a) The third–period alkali metal
(b) The third–period alkaline earth metal
(c) The rare earth element with the highest atomic mass
(d) The lanthanide that is radioactive.
Periodic Table:
Transcribed Image Text:
2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 Sr 85.47 87.62 20 38 Ca Sc 40.08 44.96 21 56 Cs La Ba 132.91 137.33 138.91 88 3 IIIB 39 Y 88.91 57 89 Atomic number Symbol Atomic mass Ra Ac (226) (227) 4 IVB 22 Ti 47.88 40 Zr 91.22 72 5 VB 23 V 50.94 41 Nb 92.91 73 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 104 105 Rf Db (261) (262) (263) Sg 106 59 Pr 140.91 91 7 VIIB Hf Ta W Re 178.49 180.95 183.85 186.21 Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 Fe Co 55.85 58.93 Os 190.2 44 45 Ru Rh 101.07 102.91 76 9 VIII 61 Pm (147) 27 108 Bh Mt Hs (262) (265) (266) 93 U NP 238.03 (237) 77 Ir 192.22 109 10 VIII 62 Sm 150.36 94 28 Ni 58.69 46 Pd 106.42 78 11 IB 12 IIB 13 IIIA 5 B 10.81 13 Al 26.98 14 15 IVA 6 C 12.01 14 Si 28.09 32 17 16 VA VIA VIIA 8 7 N 14.01 15 P 30.97 79 Pt 195.08 110 Ds 81 82 Au Hg TI Pb 196.97 200.59 204.38 207.2 111 112 113 114 Rg Cn Nh Fl (271) (272) (277) (286) (285) 29 30 31 33 As Ge Se Cu Zn Ga 63.55 65.39 69.72 72.63 74.92 78.97 16.00 16 S 32.07 34 47 51 48 49 50 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 83 Bi 208.98 115 84 Po (209) 9 F 19.00 Md (258) 17 Cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 116 Mc Ts Lv (289) (293) (294) 117 66 67 70 63 64 65 68 69 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 98 99 100 Cf Es Fm (251) (252) (257) 95 101 102 Pu Am (244) (243) Cm (247) 97 Bk (247) 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Sodium Na The thirdperiod alkali metal b ...View the full answer

Answered By

Rashul Chutani
I have been associated with the area of Computer Science for long. At my university, I have taught students various Computer Science Courses like Data Structures, Algorithms, Theory of Computation, Digital Logic, System Design, and Machine Learning. I also write answers to questions posted by students in the area of and around Computer Science.
I am highly fortunate to receive great feedback on my teaching skills that keeps me motivated. Once a student sent me an email stating that I had explained to him a concept better than his professor did.
I believe in the fact that "Teaching is the best way to learn". I am highly fascinated by the way technology nowadays is solving real-world problems and try to contribute my bit to the same.
Besides tutoring, I am a researcher at the Indian Institute of Technology. My present works are in the area of Text Summarization and Signal and Systems.
Some of my achievements include clearing JEE Advanced with an All India Rank of 306 out of 1.5 million contesting candidates and being the Department Ranker 1 at my University in the Department of Computer Science and Engineering.
I look forward to providing the best Tutoring Experience I can, to the student I teach.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The fourthperiod alkali metal (b) The fourthperiod alkaline earth metal (c) The rare...
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The semimetal in the third period (b) The semimetal in the fourth period and Group...
-
Refer to the periodic table and select the symbol of the element that fits each of the following descriptions. (a) The semimetal in the second period (b) The semimetal in the fourth period and Group...
-
For the spherical coordinate system (R, , ) in Fig. 1.6, show that: Fig 1.6 X @3 0 Ex R e2 en ee X2
-
Single Audit, Internet Case. A not-for-profit organization has hired you to conduct an audit. The audit has been requested by the organizations board of directors. A mission of the organization is to...
-
The company had 100,000 shares of common stock outstanding on January 1. In addition, as of January 1, the company had issued 500 convertible bonds ($1,000 face value, 10%). The company has no other...
-
RQMs return on equity is: (a) 26%. (b) 54%. (c) 42%. (d) 19%. RQM Corporation Selected Financial Data, December 31, 2024 Net sales $1,800,000 Cost of goods sold 1,080,000 Operating expenses 315,000...
-
Job costing; actual, normal, and variation from normal costing. Braden Brothers, Inc., is an architecture firm specializing in high-rise buildings. Its job-costing system has a single direct-cost...
-
1. (a) (2 points) Consider the following zip code: What is the check digit? 20500-0001 I (b) (2 points) Determine the digit x that makes the number 8457938x345 divisible by 9. (c) (6 points) For the...
-
According to IUPAC convention, what is the designation for each of the following groups of elements? (a) Group IA (b) Group IIIA (c) Group VA (d) Group VIIA (e) Group IB (f) Group IIIB (g) Group VB...
-
Identify the group number corresponding to each of the following families of elements. (a) Boron group (b) Oxygen group (c) Nickel group (d) Copper group.
-
There are many positive outcomes from conflict that demonstrate commitment and enthusiasm about work activities. Properly managed, conflict can be an energising and vitalising force within groups and...
-
Discuss the following statement: Real GDP has decreased for two quarters in a row; we definitively are living through a contraction.
-
How do Keynesian views on macroeconomic fluctuations differ from those of classical macroeconomists?
-
Do you think that the hourly wage (i.e., the price of labor) is a relatively flexible or a relatively sticky price? Explain why.
-
If you were a CEO or a business unit head, under what conditions would you consider moving your headquarters overseas?
-
Consider the effects of an increase in the saving rate on the United States capital-labor ratio, according to the Solow model. a) What would be the immediate effect of a saving rate increase on the...
-
The Shipbuilders Council of America in Washington, D.C., publishes data about private shipyards. Among the variables reported by this organization are the employment figures (per 1000), the number of...
-
Complete the equations for the following equilibria and calculate Keq where the Keq expression includes [HO]. Be sure to enter Keq in proper scientific notation. (a) ammonia (acting as a base) reacts...
-
Norton Co. had the following amounts related to its pension plan in 2010. Actuarial liability loss for 2010 $28,000 Unexpected asset gain for 2010 18,000 Accumulated other comprehensive income (G/L)...
-
Lahey Corp. has three defined-benefit pension plans as follows. How will Lahey report these multiple plans in its financial statements? Plan X Plan Y Plan Z Pension Assets (at Fair Value) $600,000...
-
Manno Corporation has the following information available concerning its postretirement benefit plan for 2010. Service cost $40,000 Interest cost 47,400 Actual and expected return on plan assets...
-
"Civil Law versus Common Law" Please respond to the following: Predict the long-term use of both civil and common law and explain whether you believe both have a foundation strong enough to last in...
-
Briefly describe the difference between the common law and civil law traditions. (2 marks) What are the four essential elements of a valid contract in both the common law and civil law...
-
Question 6 [15 points] Today is January 1, 2024. You are helping Enrico make 10-year cash flow projections for an IT project. The cost of financing for the project can be inferred from the following...

Study smarter with the SolutionInn App


