Refer to the periodic table and state the mass of Avogadros number of atoms for each of
Question:
Refer to the periodic table and state the mass of Avogadro’s number of atoms for each of the following nonmetals.
(a) Sulfur
(b) Helium.
Periodic Table:
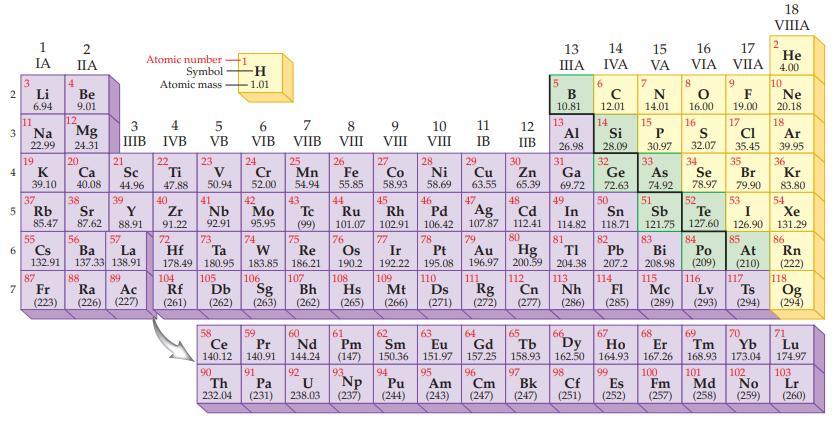
Transcribed Image Text:
19 2 3 4 5 6 7 3 11 1 IA Li 6.94 Na 22.99 19 37 4 12 87 2 IIA Fr (223) Be 9.01 Mg 24.31 20 K Ca Sc 39.10 40.08 44.96 38 21 Rb Sr Y 85.47 87.62 88.91 3 4 IIIB IVB 55 56 57 Cs Ba La 132.91 137.33 138.91 88 39 89 Atomic number Symbol Atomic mass Ra Ac (226) (227) 22 Ti 47.88 40 Zr 91.22 5 VB 23 50.94 41 Nb 92.91 -H 1.01 104 105 ᎠᏏ Rf (261) (262) 6 VIB 24 Cr 52.00 42 Mo 95.95 72 73 W Hf Ta 178.49 180.95 183.85 74 106 Sg (263) 58 Ce 140.12 90 Th Pa 232.04 (231) 59 Pr 140.91 91 7 VIIB 25 Mn 54.94 43 Tc (99) 75 Re 186.21 107 8 VIII 60 Nd 144.24 92 26 Fe 55.85 44 76 77 Ir 192.22 109 Bh Hs Mt (262) (265) (266) Os 190.2 Ru Rh 101.07 102.91 108 61 Pm (147) 9 VIII 93 27 U Np 238.03 (237) Co 58.93 45 10 VIII 28 94 Ni 58.69 63 62 Sm 150.36 151.97 Eu 95 Am Pu (244) (243) 64 Gd 157,25 96 Cm 13 IIIA (247) 5 B 10.81 65 Tb 158.93 97 Bk (247) 13 14 IVA 6 11 12 Al Si IB IIB 26.98 28.09 с 12.01 14 7 15 VA N 14.01 29 Cu 63.55 47 46 48 52 Pd 106.42 78 79 83 85 49 50 51 53 Ag Cd In Sn Sb Te I 107.87 112.41 114.82 118.71 121.75 127.60 126.90 80 81 82 84 Au Pb 196.97 200.59 204.38 207.2 110. 111 112 113 114 Ds Rg Cn Nh FI (271) (272) (277) (286) (285) Pt 195.08 Hg ΤΙ Bi Po At 208.98 (209) (210) 115 116 117 Mc Lv Ts (289) (293) (294) 16 VIA 8 16.00 15 16 P S 30.97 32.07 33 34 As Se 30 31 32 35 Zn Ga Ge Br 65.39 69.72 72.63 74.92 78.97 79.90 17 VIIA 9 F 19.00 17 Cl 35.45 66 67 68 69 70 Dy Ho Er Tm Yb 162.50 164.93 167.26 168.93 173.04 99 100 101 102 Cf Es Fm Md (252) (257) (258) 98 No (251) (259) 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118. Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a 3...View the full answer

Answered By

Kainat Shabbir
i am an experienced qualified expert with a long record of success helping clients overcome specific difficulties in information technology, business and arts greatly increasing their confidence in these topics. i am providing professional services in following concerns research papers, term papers, dissertation writing, book reports, biography writing, proofreading, editing, article critique, book review, coursework, c++, java, bootstarp, database.
5.00+
184+ Reviews
255+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and state the mass of 6.02 x 10 23 atoms of each of the following nonmetals. (a) Beryllium (b) Barium (c) Boron (d) Bromine. 2 3 4 AD 6 3 7 11 1 IA 37 5 Rb 4 Li B Na 19 55...
-
Refer to the periodic table and state the mass of 6.02 x 10 23 atoms of each of the following metals. (a) Sodium (b) Strontium (c) Silicon (d) Selenium. 2 3 4 av 6 3 7 11 1 IA 37 5 Rb 4 Li B Na 19 55...
-
Refer to the periodic table and state the mass for each of the following number of atoms. (a) 1 atom of carbon (b) 6.02 x 10 23 atoms of carbon. 2 3 4 av 6 3 7 11 1 IA 37 5 Rb Li Na 19 55 87 Fr 4 2...
-
In Problem perform the indicated operations and reduce answers to lowest terms. 2 4 ? 16 x? + 4x
-
Warwick Online Company has the following liability accounts after posting adjusting entries: Accounts Payable $63,000, Unearned Ticket Revenue $24,000, Estimated Warranty Liability $18,000, Interest...
-
The crossbar wrench is used to remove a lug nut from the automobile wheel. The mechanic applies a couple to the wrench such that his hands are a constant distance apart. Is it necessary that a = b in...
-
Consider a square loop of wire that carries a clockwise current when viewed from above. What is the direction of the magnetic field at the center of the square loop due to \((a)\) the left side,...
-
VolWorld Communications Inc., a large telecommunications company, is evaluating the possible acquisition of Bulldog Cable Company (BCC), a regional cable company. VolWorld's analysts project the...
-
1. 49 The value of n(n+n+1) is equal to (10a) [10a-3. The value of (a + ) is n=1 48 47 46 2 1 2. If + + + + + = (2)(3) (3)(4) (4)(5) (48)(49) (49)+(50) k is equal to 3. +++++ Then 4. 5. In a...
-
Refer to the periodic table inside the front cover of this textbook. State the mass of Avogadros number of atoms for each of the following metals. (a) Silver (b) Mercury Periodic Table: 2 3 4 5 6 7 3...
-
Balance each of the following combustion reactions by inspection. (a) CH 4 (g) + O 2 (g) CO 2 (g) + H 2 O(g) (b) C 3 H 8 (g) + O 2 (g) S CO 2 (g) + H 2 O(g).
-
The following selected transactions were completed by Minty Supplies Co., which sells irrigation supplies primarily to wholesalers and occasionally to retail customers: Jul. 5. Sold merchandise on...
-
If you sold it for 4,000,000 the investment 6 years from now and received the $100,000 cash flow each of the next 6 years, what would your compound annual return actually be on this investment?
-
2). King Tut ruled ancient Egypt from 1332 to 1323 B.C., but died as a teenager. An analysis of STR markers carried out on samples from the mummies of King Tut and several other mummies found in...
-
please provide and detail specific routes the carriers work on, do they provide mainline routes, regional services, feeder services etc. please research and discuss if you are not researching a...
-
What are other thesis papers on investment scams on the field of financial management?
-
Explain the differences between capital expenditure and capital budgeting. Provide examples for each within the context of a hospital environment. Discuss how they relate to each othe?
-
Tim Waters, the COO of BioDerm, has asked his cost management team for a product-line profitability analysis for his firms two products, Xderm and Yderm. The two skin care products require a large...
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
Accounting Change Presented are income statements prepared on a LIFO and FIFO basis for Carlton Company, which started operations on January 1, 2009. The company presently uses the LIFO method of...
-
Accounting Changes Depreciation Robillard Inc. acquired the following assets in January of 2007. Equipment estimated service life, 5 years; salvage value , $15,000 $465,000 Building, estimated...
-
Change in Estimate and Error; Financial Statements Presented below are the comparative income statements for Pannebecker Inc. for the years 2009 and 2010. The following additional information is...
-
A single nuclear reactor produces 2.6GW of electrical power, and has a generator voltage of 22kV.What percentage of power would be lost from our nuclear reactor in three-line transmission that goes...
-
6. A carver begins work on a block of granite that measures 20 cm by 10 cm by 5 cm. Granite has a density of 2.7 g/cm. What is the mass of the piece of granite? a. What do you need to calculate...
-
3. The distance between carbon atoms in diamond is 0.154 nm. What is this distance in meters? Have Factor 0.154 nm 1 x 10^9nm Want 1.54 x 10^-10m 4. Calculate the number of grams in 32.0 lbs....

Study smarter with the SolutionInn App


