Refer to the periodic table on the inside cover of this text and determine the atomic number
Question:
Refer to the periodic table on the inside cover of this text and determine the atomic number and mass number for element 92, uranium.
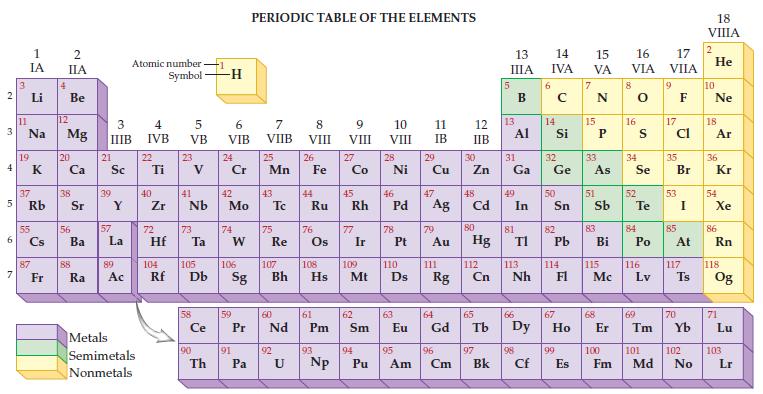
Transcribed Image Text:
2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -1 Th -H 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 Re 60 P Pr Nd 92 8 VIII U 26 44 Fe Ru 76 61 Pm 93 9 VIII 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Np 27 Co 45 Rh 77 Ir 62 Sm 94 10 VIII Pu 28 Ni 46 78 47 Pd Ag 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 31 Ga Al 49 81 In 66 TI 113 Nh 98 Cf 6 14 15 16 IVA VA VIA 14 Si 32 Ge 50 82 Pb 114 67 99 E Es 15 33 As 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 Cl 35 53 85 Br I At 117 70 Ts 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
In the periodic table we observe The atomic n...View the full answer

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table on the inside cover of this text and determine the atomic number and atomic mass for iron. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra...
-
Determine the atomic number Z, the atomic mass number A, and the element X for the unknown species in the following reaction for the fission of uranium : Consult the periodic table on the inside of...
-
Refer to the periodic table on the inside front cover and indicate (a) The most nonmetallic element; (b) The transition metal with lowest atomic number; (c) A metalloid whose atomic number is exactly...
-
The following two equations have a common solution of (1, 2, 3). Which equation would complete a system of three linear equations in three variables having solution set {(1, 2, 3)}? x+y+z=6 2x = y +...
-
How do product costs affect the financial statements? How does the classification of product cost (as an asset vs. an expense) affect net income?
-
At week 24 of a project to shoot a television commercial, what should the expenditures be? If the earned value is right on schedule but the actual expenses are $9,000, what are the cost and schedule...
-
Taking off on a runway, a small jet experiences a forward force of \(90,000 \mathrm{~N}\) exerted by the engine and a backward force of \(16,000 \mathrm{~N}\) exerted by the air. Choose a system and...
-
Consider a competitive market served by many domestic and foreign firms. The domestic demand for these firms product is Qd 600 2P. The supply function of the domestic firms is QSD = 200 P, while that...
-
You have saved $500 to buy an electric scooter that costs $1800. You plan to borrow the rest of the purchase price from the bank. The bank is offering a 3 year loan at 4% APR with monthly payments....
-
Refer to the periodic table and determine the atomic number and mass number listed for element 61, promethium. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB...
-
How many neutrons are in the nucleus of an atom of cobalt-60? (a) 27 (b) 33 (c) 60 (d) 87 (e) None of the above.
-
Given the following AOA network, determine the first activity to be given extra resources by the following priority rules: (a) Shortest task first (b) Minimum slack first (c) Most critical followers...
-
Why does the Joint Commission now require hospitals and other healthcare organizations to have a performance management system?
-
In designing a benefits plan, what are the most important considerations for an employer?
-
Mapleton Family Medicine is a physician group practice located in a small city (population 150,000) in the Midwest. Mapleton is an eight-physician practice, consisting of family physicians,...
-
Why are there are no such consequences to patients who demand doctors, nurses, or workers of a specific race to administer their healthcare?
-
What are some of the behaviors that may indicate to managers that organizing activities are occurring?
-
Explain why you believe that the courts have found each of the following to either constitute or not constitute a regulatory taking: (a) A city ordinance reducing the size limit for freestanding...
-
Suppose that a business sells 6-month subscriptions to its monthly magazine. On January 1, the company receives a total of $600 for 10 subscriptions. To record this transaction, the company debits...
-
What are holding companies? What are their advantages and disadvantages?
-
Discuss, in general, what it means for the brothers to set a credit and collections policy.
-
What is the firms expected days sales outstanding (DSO)?
-
The table shows the demand schedule facing Nina, a monopolist selling baskets. What is the change in total revenue if she raises the price from $10 to $12? Price Number of Baskets Sold $ 20 3 18 5 16...
-
When neither residents nor nonresidents are allowed to convert a currency to a foreign currency, the currency is considered Multiple choice question. nonconvertible. freely convertible. internally...
-
Assume that the typical consumer basket consists of 5 bushels of peaches and 10 bushels of walnuts and that the base year is 2014. What is the consumer price index for 2014? Year peaches pecans 2014...

Study smarter with the SolutionInn App


