Refer to the periodic table on the inside cover of this text and determine the atomic number
Question:
Refer to the periodic table on the inside cover of this text and determine the atomic number and atomic mass for iron.
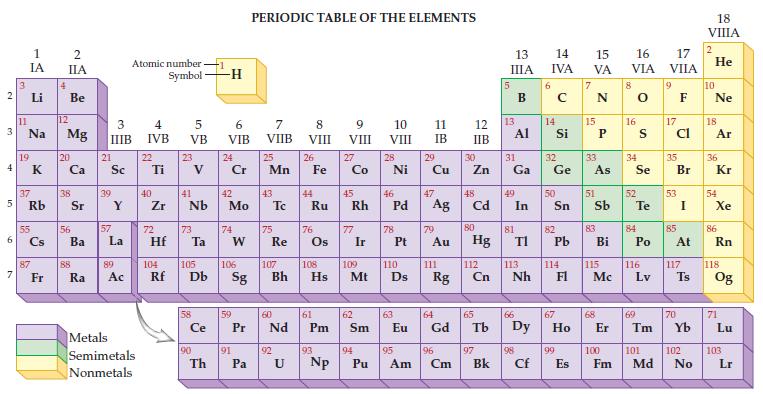
Transcribed Image Text:
2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac Atomic number Symbol Metals Semimetals Nonmetals 4 IVB 22 Ti 40 Zr 5 VB 72 23 41 Nb 73 Hf Ta Ce 90 -1 Th -H 6 VIB 24 Cr 42 Mo 74 W 59 91 PERIODIC TABLE OF THE ELEMENTS 7 VIIB Pa 25 Mn 43 75 Re 60 P Pr Nd 92 8 VIII U 26 44 Fe Ru 76 61 Pm 93 9 VIII 104 105 106 107 108 109 110 Rf Db Sg Bh Hs Mt D Np 27 Co 45 Rh 77 Ir 62 Sm 94 10 VIII Pu 28 Ni 46 78 47 Pd Ag 63 11 IB 29 95 Cu 79 Au 111 64 Eu Gd Rg 96 Am Cm 12 IIB 30 Zn 48 Cd 80 Hg 112 Cn 65 97 Bk 13 IIIA 13 31 Ga Al 49 81 In 66 TI 113 Nh 98 Cf 6 14 15 16 IVA VA VIA 14 Si 32 Ge 50 82 Pb 114 67 99 E Es 15 33 As 51 83 Bi 115 Mc 68 8 16 34 Se 52 S Te 84 Po 116 Lv 69 17 VIIA 9 17 Cl 35 53 85 Br I At 117 70 Ts 100 101 102 Fm Md No 18 VIIIA 2 He 10 Ne 18 Ar 36 Kr 54 Xe 86 Rn 118 Og 71 Lu 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
In the periodic table we observe The atom...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table on the inside cover of this text and determine the atomic number and mass number for element 92, uranium. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20...
-
Refer to the periodic table on the inside front cover and indicate (a) The most nonmetallic element; (b) The transition metal with lowest atomic number; (c) A metalloid whose atomic number is exactly...
-
Refer only to the periodic table on the inside front cover and indicate which of the atoms, Bi, S, Ba, As, and Ca, (a) Is most metallic; (b) Is most nonmetallic; (c) Has the intermediate value when...
-
Write using only positive exponents and then evaluate. Assume that all variables represent nonzero real numbers. 5 -2
-
What does the value-added principle mean as it applies to managerial accounting information? Give an example of value added information that may be included in managerial accounting reports but is...
-
Which of the four examples of conflict resolution is the best example, in your opinion, of effective resolution? Why?
-
When you stand up from a seated position, you push down with your legs. Does this mean you do negative work when you stand up?
-
The following transactions of Weber Company occurred during 2016: 1. The company acquired a tract of land in exchange for 1,000 shares of $ 10 par value common stock. The stock was traded on the New...
-
What is the sharpe ratio (annualized) when given a risk-free rate of 0.0, an average daily return of 0.003, a standard deviation of daily returns of 0.01, and 75 days of daily data.
-
Refer to the periodic table and determine the atomic number and atomic mass for sulfur. 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2 IIA Be 12 Mg 20 38 56 Ba 88 Ra 3 IIIB 21 Sc 39 57 Y La 89 Ac...
-
Calculate the atomic mass of copper given the following data: Isotope 63 Cu 65Cu Mass 62.930 amu 64.928 amu Abundance 69.09% 30.91%
-
The company operates a travel club through which subscribers can access low rates for air fares, hotel rooms, and rental cars. Each year, subscribers pay a refundable fee of $2,000 that allows them...
-
Redraft the following cash flow statement as per the provisions of accounting standard (AS-3) applicable in India: Particulars Net profit (before interest, tax and dividend) Add: Sales of investments...
-
What is the publics perception of healthcare institutions as being in a perpetual state of disaster preparedness? How does this view differ from the number of injuries and illnesses reported in...
-
How is organizational philosophy reflected in the measurement of nurse workload?
-
Think about your own experience or that of a family member in receiving healthcare services. To what degree was the healthcare provider customeroriented? Why? What lessons can you derive from that...
-
Describe and discuss future-oriented job analysis and generic job analysis. How may each be used to help healthcare executives cope with a rapidly changing and competitive environment? What are some...
-
Over the past decade, a number of groups and individuals have argued against many of the legal protections that corporations receive. These critics argue that problems such as those associated with...
-
Which of the following statements is false? a. Capital leases are not commonly reported in a Capital Projects Fund. b. A governmental entity may report a Capital Project Fund in one year but not the...
-
What is the dollar amount of the firm's current bad debt losses? What losses would be expected under the new policy?
-
What would be the firm's expected dollar cost of granting discounts under the new policy?
-
What is the firm's current dollar cost of carrying receivables? What would it be after the proposed change?
-
Regions Clip Hospital, Corp. specializes in providing patients with novel therapeutic treatments for cancer. They've made some investment related decisions over the past couple of years, including...
-
Go to at least three of the financial websites listed under: Yahoo! Financial section (http://biz.yahoo.com/ne.html) Google Financial section (http://www.google.com/finance) Morningstar...
-
1. You have a saving account that you were told has an effective annual return of 9% and compounds daily. What is the Annual Percentage Rate you on this account? 2. You purchased a stock six months...

Study smarter with the SolutionInn App


