The compound zirconium silicate occurs in cubic zirconia. Use the periodic table to predict the formula for
Question:
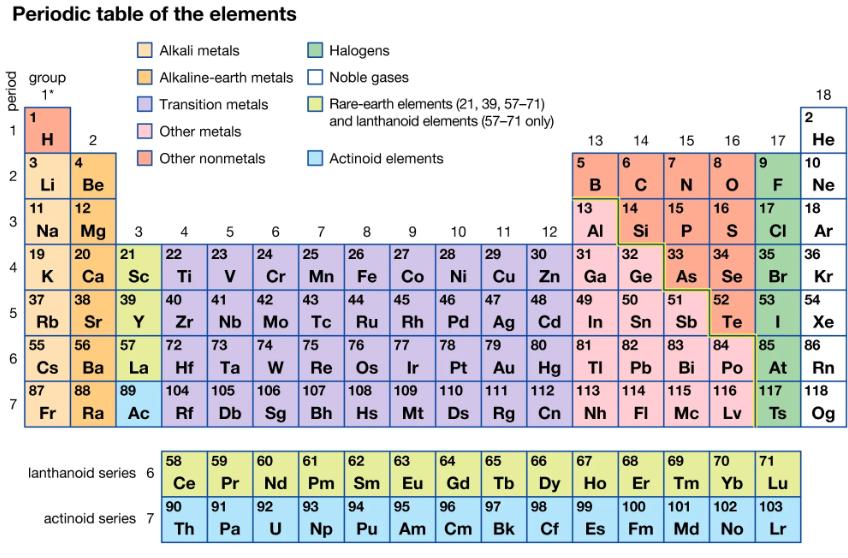
The compound zirconium silicate occurs in cubic zirconia. Use the periodic table to predict the formula for zirconium silicate, given the formula of titanium silicate, TiSiO4.
Transcribed Image Text:
Periodic table of the elements Alkali metals Alkaline-earth metals Transition metals Other metals Other nonmetals period - 2 3 4 5 6 7 group 1* 3 H 2 4 Li 11 Na 19 K 37 Rb 55 Cs 87 Fr Be 12 Mg 20 21 22 23 Ca Sc Ti V 38 39 40 41 Sr Y Zr 56 57 72 73 74 Ba La Hf Ta W 88 89 104 105 Ra Ac Rf 3 lanthanoid series 6 actinoid series 7 4 58 Ce 90 Th 5 es Db Sg 7 Halogens Noble gases Pa U Rare-earth elements (21, 39, 57-71) and lanthanoid elements (57-71 only) Actinoid elements 8 9 6 24 25 26 28 Cr Mn Fe Co Ni 45 47 48 Ag Cd Sn Sb 79 80 81 82 83 42 43 44 46 Nb Mo Tc Ru Rh Pd 75 76 77 78 Re Os Ir Pt Au Hg TI Pb Bi 107 108 109 110 111 112 113 114 Bh Hs Mt Ds Rg Cn Nh FI 27 59 60 61 62 63 Pr Nd Pm Sm Eu 91 92 93 94 95 Np Pu Am 10 64 Gd 96 Cm 11 29 30 Cu Zn 12 97 Bk 13 5 6 B 14 65 66 67 Tb C 49 14 31 32 Ga Ge 50 7 13 15 Al Si P 33 34 35 As Se Br 68 15 Dy Ho Er 98 99 100 Cf Es Fm N 16 17 8 OF 16 S 84 9 10 18 2 Po At 115 116 117 Mc Lv Ts He 17 18 CI 69 70 71 Tm Yb Lu 101 102 103 Md No Lr Ne Ar 51 52 53 54 Te I 85 86 36 Kr Xe Rn 118 Og
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
Formula for zirconium silicate ZrSiO can be predicted based on the formula for titanium silicate TiS...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
The compound titanium oxide occurs in the mineral rutile. Use the periodic table to predict the formula for titanium oxide, given the formula of zirconium oxide, ZrO 2 . Periodic table of the...
-
The element actinium is unstable and radioactive. Use the periodic table to predict the formula for actinium chloride, given the formula of lanthanum chloride, LaCl 3 . Periodic table of the elements...
-
The element lawrencium is unstable and radioactive. Use the periodic table to predict the chemical formula for lawrencium chloride, given the formula of lutetium chloride, LuCl 3 .
-
A sealed flask contains water and oxygen gas at 25C. The O 2 gas has a partial pressure of 1.5 atm. (a) What is the concentration of O 2 in the water? (b) If the pressure of O 2 in the flask is...
-
Recording Adopted Budget. The City of Marion adopted the following General Fund budget for fiscal year 2011: Estimated revenues: Taxes................$3,000,000 Intergovernmental revenues...........
-
Using the chain ratio method described in Appendix 2, estimate the market potential for the Fisker Karma sports car. Search the Internet for reasonable numbers to represent the factors you identified...
-
a. Is a \(4 p ightarrow 4 s\) transition allowed in sodium? If so, what is its wavelength? If not, why not? b. Is a \(3 d ightarrow 4 s\) transition allowed in sodium? If so, what is its wavelength?...
-
A regression analysis of data on n = 53 males considered the following variables: Y = SBPSL (estimated slope based on the straight-line regression of an individual's blood pressure over time) X1 =...
-
Advantages and disadvantages of share buybacks vs dividend payments, for both the investor and manager.
-
Supply a systematic name for each of the following binary ionic compounds: (a) Mn 3 P 2 (b) Fe 2 S 3 .
-
Classify each of the following as a binary ionic compound, ternary ionic compound, binary molecular compound, binary acid, or ternary oxyacid: (a) Carbon disulfide, CS 2 (b) Lithium dichromate, Li 2...
-
Assume Polaris invested $ 2.12 million to expand its manufacturing capacity. Assume that these assets have a ten-year life, and that Polaris requires a 10% internal rate of return on these assets....
-
A steel service center sources products from an integrated steel mill at a cost of $2,000 per ton. Demand for steel at the service center is 50 tons per month. The service center has an annual...
-
The forecasting function is the foundation of any revenue management system. unnecessary for a revenue management system. an added plus for any revenue management system. likely to create problems...
-
Offering a combination of which of these goods is a form of revenue management? DVDs and streaming services Extra crispy and original recipe fried chicken Paperback and hardcover books Rolls Royce...
-
Which is an example of balancing peak-load demand? Car dealers offer rebates in July and August, just before the next years models are shipped to their lots. An assistant dean signs all of the course...
-
What are the major steps required in implementing sales and operations planning in supply chain management?
-
Test the following sequence of observations by using the runs test and = .05 to determine whether the process produced random results. X X X Y X X Y Y Y X Y X Y X X Y Y Y Y X
-
What is your assessment of the negotiations process, given what you have studied? What are your recommendations for Mr. Reed? You must justify your conclusions
-
Seles Corporations charter authorized issuance of 100,000 shares of $10 par value common stock and 50,000 shares of $50 preferred stock. The following transactions involving the issuance of shares of...
-
Before Gordon Corporation engages in the treasury stock transactions listed below, its general ledger reflects, among others, the following account balances (par value of its stock is $30 per share)....
-
(Treasury StockCost MethodEquity Section Preparation) Washington Company has the following stockholders equity accounts at December 31, 2010. Common Stock$100 par value, authorized 8,000...
-
4. An artist company prints and sells x copies of one of her most popular original paintings every week. The weekly price-demand and cost equations are, respectively, = 400 - 0.4x and C(x) = 2,000 +...
-
1.In 2017, the Tax Cuts and Jobs Act (TCJA) was passed. Evaluate and share the differences in the concepts and principles of this federal tax law compared to the old federal tax law. 2. Using...
-
Use the substitution method to find the following integral x dx u = x + 3 (x + 3)4

Study smarter with the SolutionInn App


