Find the modulus and argument of (3 + j4)*(12 - j5) (3 - j4)(12 + j5)
Question:
Find the modulus and argument of
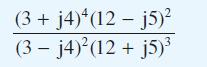
Transcribed Image Text:
(3 + j4)*(12 - j5) (3 - j4)(12 + j5)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
3j4 12j52 34 12 j5 3j4412j5 3j...View the full answer

Answered By

Dinesh F
I have over 3 years of professional experience as an assignment tutor, and 1 year as a tutor trainee.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
can you help explain the 1975 Securities Exchange Commission violation by Gulf Oil Corporation?
-
1. Find the modulus and argument of (1 + i)^6. 2. Find the roots of the equation z^3 - 2z^2 + (2 + 2i)z - (1 + 3i) = 0. 3. Find the complex number w such that w^4 = (1 + i)^4 and w has a negative...
-
a. Find the exact values of the modulus and argument of z. b. Given that Write z/w in the form re i , where > 0 and < < . z = 4/3 4i w = 22 COS +isin 12 I 12
-
Lead nitrate solution is added to a test tube containing potassium iodide solution. (a) Write the name and colour of the compound precipitated. (b) Write the balanced chemical equation for the...
-
Describe how the big five personality attributes might affect a managers own behavior in dealing with subordinates.
-
M. Gonzales has the following transactions during August of the current year. Indicate (a) The effect on the accounting equation (b) The debit-credit analysis illustrated on pages 6165 of the...
-
What were two of the most profound influences in the development of present-day hospitals?
-
Examine and compare one of the following product sets. Base your comparison on such factors as features, costs, convenience, ease of use, and value. a. GPS versus maps. b. Cell phones versus...
-
A particle's position is given by x = 2 - 11t + 2t2 (where t is in seconds and x is in meters). (a) What is its velocity at t = 1 s? (Indicate the direction with the sign of your answer.) -7 m/s (b)...
-
Cyclohexanol was passed over a catalyst to form water and cyclohexene: CyclohexanolcatalystWater+Cyclohexence It is suspected that the reaction may involve a dual-site mechanism, but it is not known...
-
Experimental data for the gas-phase catalytic reaction A + B C is shown below. The limiting step in the reaction is known to be irreversible, so that the overall reaction is irreversible. The...
-
Write a function that has this prototype: int replace(char * str, char c1, char c2); Have the function replace every occurrence of c1 in the string str with c2, and have the function return the...
-
Date Information 13/05/20XX BAC Commercial pays monthly rent of $2000 by EFT. 15/05/20XX The tenant (renter) Jill Sharp pays by EFT the rental of $1350 for 15 Drysdale Ave. 20/05/20XX Fred Sykes...
-
How will attending the DVAA benefit your agency or department as a whole?
-
Use the above information to complete the following tasks: Prepare a cash budget for the months of January 2022, February 2022, March 2022 and April 2022. Based on the cash budget, provide...
-
What happens if an organization does not have the three sections listed on their statement of cash flows?
-
You decided to create a new language! Your computer memory has 4 bits, so what is the Maximum number of symbols you can encode in this memory?
-
When adding real estate to an asset allocation program that currently includes only stocks, bonds, and cash, which of the properties of real estate returns affect portfolio risk? Explain. a. Standard...
-
At Glass Company, materials are added at the beginning of the process and conversion costs are added uniformly. Work in process, beginning: Number of units Transferred - in costs Direct materials...
-
Count the total number of s bonds and p bonds in the compound below: -3-N
-
Calculate the standard enthalpy of formation of FeS 2 (s) at 600. °C from the following data at 298.15 K. Assume that the heat capacities are independent of temperature. You are also given that...
-
Use the tabulated values of the enthalpy of combustion of benzene and the enthalpies of formation of CO 2 (g) and H 2 O(l) to determine H o f for benzene.
-
Why are Reserves created by companies? Distinguish between (i) Free Reserves and 'Specific Reserve' and (ii) Revenue Reserve and Capital Reserve.
-
What are the basic philosophies of construction management? (in depth)
-
Suppose you can borrow and lend at an annual interest rate of 6% per annum. The IBM stock is trading at $120. It is not going to pay any dividend in one year. Ignore all transaction costs. (a) What...

Study smarter with the SolutionInn App


