If y = u(x)v(x), prove that Hence prove Leibnizs theorem for the nth derivative of a product:
Question:
If y = u(x)v(x), prove that
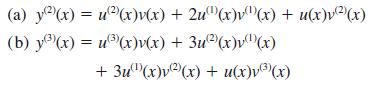
Hence prove Leibniz’s theorem for the nth derivative of a product:
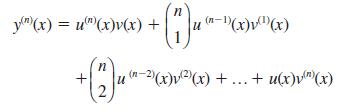
Transcribed Image Text:
(a) y(x) = u(x)v(x) + 2u(x)v(x) + u(x)v)(x) (b) y(x) = u(x)v(x) + 3u2(x)v(x) + 3u(x)v(x) + u(x)v(x)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a b y uxvx...View the full answer

Answered By

Keziah Thiga
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagirism), well-researched and critically analyzed papers.
4.90+
1504+ Reviews
2898+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use Leibnizs theorem (Question 71) to find the following: Data from Question 71 If y = u(x)v(x), prove that Hence prove Leibnizs theorem for the nth derivative of a product: (a) d dxs (x sin x) (put...
-
One of several Leibniz Rules in calculus deals with higher-order derivatives of products. Let (fg) (n) denote the nth derivative of the product fg, for n 1. a. Prove that (fg) (2) = f"g + 2 f'g' +...
-
Suppose the European Parliament is considering legislation that will decrease tariffs on agricultural products imported from the United States. The benefit of the tariff reduction is estimated at 5...
-
Find the directive gain and directivity of the small loop antenna.
-
A steel tank contains 300 g of ammonia gas (NH3) at a pressure of 1.35 x 106 Pa and a temperature of 77oC. (a) What is the volume of the tank in liters? (b) Later the temperature is 22oC and the...
-
What is the specificity of the test? Mental Health The Chinese Mini-Mental Status Test (CMMS) consists of 114 items intended to identify people with Alzheimers disease and senile dementia among...
-
Question: Kelly Hackworth took a leave of absence from her job at Progressive Insurance, to care for her ailing mother. When she offered to return, Progressive refused to give her the same job or one...
-
The Fish House (TFH) in Norfolk, Virginia, sells fresh fish and seafood. TFH receives daily shipments of farm-raised trout from a nearby supplier. Each trout costs $2.45 and is sold for $3.95. To...
-
Paul, is employed by PO Advocates, a firm of practicing lawyer. He made the following payments during the year 2019. i) Mortgage interest of shs 120,000 on a shs 850,000 mortgage provided by Housing...
-
Find the radius of curvature at the point (2, 8) on the curve y = x 3 .
-
Prove that where d" - (e sin bx) = (a + b)/ex sin(bx + n0) d.x"
-
The random variable X, representing the number of errors per 100 lines of software code, has the following probabilitydistribution: 3 0.01 4 0.3 0.25 0.4 0.04
-
What format requirements must be met?
-
What needs to be contained in the caption of a complaint?
-
How to draft a statement of fact for complaints?
-
What the general requirements are for all pleadings?
-
What are the possible consequences for failing to file the civil cover sheet?
-
How can social software enhance communication?
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Consider the following mechanism, which results in the formation of product P: If only the species A is present at t = 0, what is the expression for the concentration of P as a function of time? You...
-
Using the preequilibrium approximation, derive the predicted rate law expression for the following mechanism: A+B- P
-
For the reaction I (aq) + OCl (aq) OI (aq) + Cl (aq) occurring in aqueous solution, the following mechanism has been proposed: a. Derive the rate law expression for this reaction based on this...
-
8 Under Hitler's leadership, Germany formally announced a projected goal of having an army of the following size (pick one): a) 16 divisions, after careful analysis concluded that Germany lacked...
-
A credit balance in Retained Earnings is normal, indicating that the corporation's lifetime earnings exceed lifetime losses and dividends. Question 11 options: True False Question 12 (1 point) Saved...
-
If an investor does not diversify his portfolio and instead puts all of his money in one stock, the appropriate measure of security risk for that investor is the ________. If the investor considers...

Study smarter with the SolutionInn App


