Consider the following mechanism, which results in the formation of product P: If only the species A
Question:
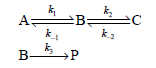
If only the species A is present at t = 0, what is the expression for the concentration of P as a function of time? You can apply the preequilibrium approximation in deriving your answer.
Transcribed Image Text:
k, ec B: A: k1 ka →P B-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Using the preequilibrium approximation we can express B and C ...View the full answer

Answered By

Omar ELmoursi
I'm Omar, I have Bachelor degree in Business and Finance, My unique approach is to help students with questions and assignments, I can teach Business, Math, Accounting, Managerial Accounting, Economy, Human resources management, organizational behavior, project management, I have experience dealing with different types of students and teach them how to deal with different types of exercises.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the following mechanism for renaturation of a double helix from its strands A and B: Derive the rate equation for the formation of the double helix and express the rate constant of the...
-
Consider the following mechanism for the thermal decomposition ofR2: Where R2, PA' PB are stable hydrocarbons and Rand R' are radicals. Find the dependence of the rate of decomposition of R, on the...
-
Consider the following mechanism for a reaction in aqueous solution and indicate the species acting as a catalyst: Explain why you believe this species is a catalyst. What is the overall reaction?...
-
Question #1 The following table indicates the net cash flows of a capital asset: Year Net Cash Flow 0 $-8,400 1 $5,900 2 $6,200 Do not enter dollar signs or commas in the input boxes. Use the...
-
Determine whether the statement is true or false. Justify your answer. 1. If f is an even function, then f1 exists. 2. If the inverse function of f exists and the graph of f has a y-intercept, then...
-
What are your personal growth objectives for your venture? Is a lifestyle business going to meet your personal goals? Or a highpotential venture?
-
The internal energy of an ideal gas is a function of (a) Temperature only (b) Pressure only (c) Volume only (d) All of these.
-
Ann Donnelly is a senior audit manager in an East Coast office of a public accounting firm. Her prospects for promotion to partner are excellent if she continues to perform at the same high-quality...
-
Pn Noraini owned a property and provide the following information for the year ended 31 December 2015: Receipts: RM Rental: Jan - Dec 2015 40000 Jan - Mar 2016, received in Dec 2015 10000 Deposits...
-
Strong Shoes' comparative balance sheet is presented below. Strong reports under ASPE. Additional information: 1. Profit was $28,300. Dividends declared and paid were $26,400. 2. Equipment that cost...
-
Consider the gas-phase isomerization of cyclopropane. Are the following data of the observed rate constant as a function of pressure consistent with the Lindemann mechanism? CH, CH,CH=CH2 CH,-CH2 k...
-
Using the preequilibrium approximation, derive the predicted rate law expression for the following mechanism: A+B- P
-
Who can perform work that may be of value to the independent auditor?
-
escribe how the three-dimensional structure of an enzyme determines its specificity and catalytic mechanism. How do techniques like X-ray crystallography and cryo-electron microscopy contribute to...
-
Ann Parsons deposits $10,000 in a savings account each year earning 6% interest annually (EOY). (i) How much will be in the account at the end of the 15th year? (ii) How much would you have if Ann...
-
What are the key differences between competitive, non-competitive, and uncompetitive enzyme inhibition? How are these concepts applied in the development of enzyme-based therapeutics ?
-
KFC Beef, a fast-food company selling roasted beef in outlets through the country, went public in 1993. In the year prior to going public, it had revenues of $40 million, on which it reported EBIT of...
-
Mike's Auto Parts and Repairs Service's income statement data for the year ended August 31, 2018, follow. (Click the icon to view the data.) Assume the ending inventory was accidentally overstated by...
-
In how many ways can 2 teams from 14 teams in the American League be chosen without regard to which team is at home?
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
a. Calculate S if 1.00 mol of liquid water is heated from 0.00C to 10.0C under constant pressure if C P,m = 75.3 J K -1 mol -1 . b. The melting point of water at the pressure of interest is 0.00C and...
-
Show what reagents you would use to prepare each of the following ethers via an alkoxymercuration-demercuration. a. b. c. d. OEt
-
A 2.25 mole sample of carbon dioxide, for which C P,m = 37.1 J K -1 mol -1 at 298 K, is expanded reversibly and adiabatically from a volume of 4.50 L and a temperature of 298 K to a final volume of...
-
Assume you are a trader with JP Morgan. From the quote screen on your computer terminal, you notice that Bank A is quoting 0.8354/$1.00 and Bank Bis offering SF1.0913/$1.00. You learn that Bank Cis...
-
Leven Clinic uses client-visits as its measure of activity. During September, the clinic budgeted for 3,000 client-visits, but its actual level of activity was 3,050 client-visits. The clinic has...
-
If 2.25 grams of calcium hydroxide is dissolved in 154.56 ml of water, a. What's the Molarity (M), if the volume of solution is 500.00 ml? b. What's the Molarity (M) of hydroxide ions, if the volume...

Study smarter with the SolutionInn App


