Assign the H 2 pure rotational transitions in Figure 8.8, using data from Table 9.2 to calculate
Question:
Assign the H2 pure rotational transitions in Figure 8.8, using data from Table 9.2 to calculate the predicted frequencies. Assuming the spectra were taken at room temperature, account for the relative intensities of each transition.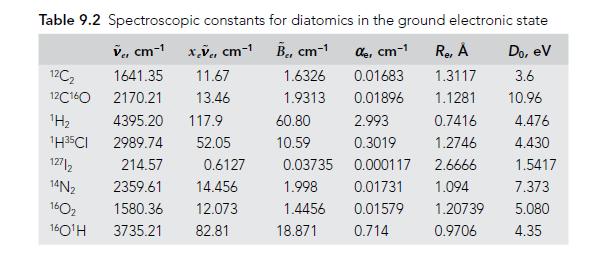
Figure 5.8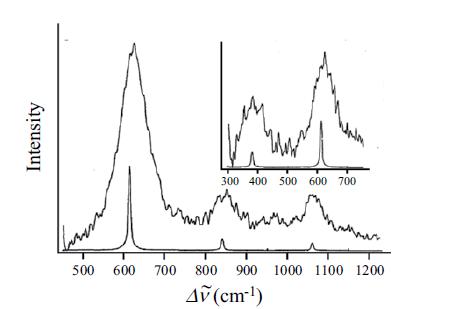
Transcribed Image Text:
Table 9.2 Spectroscopic constants for diatomics in the ground electronic state Ver cm-¹x.ve, cm-¹ B, cm-¹ de, cm-1 1.6326 0.01683 1.9313 0.01896 12C₂ 12C160 1641.35 2170.21 ¹H₂ 1H35CI 12712 14N₂ 1602 160¹H 3735.21 11.67 13.46 4395.20 117.9 2989.74 52.05 214.57 2359.61 14.456 1580.36 12.073 82.81 0.6127 60.80 10.59 0.03735 1.998 1.4456 18.871 R₂, Å 1.3117 1.1281 2.993 0.7416 0.3019 1.2746 0.000117 2.6666 0.01731 1.094 0.01579 0.714 1.20739 0.9706 Do, ev 3.6 10.96 4.476 4.430 1.5417 7.373 5.080 4.35
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Using the information from Table 92 we can assign the pure rotational transitions for H2 First we mu...View the full answer

Answered By

Allan Olal
I have vast tutoring experience of more than 8 years and my primary objective as a tutor is to ensure that a student achieves their academic goals.
4.70+
78+ Reviews
410+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using data from Table 13.3, calculate the freezing and boiling points of each of the following solutions: (a) 0.22 m glycerol (C3H8O3) in ethanol, (b) 0.240 mol of naphthalene (C10H8) in 2.45 mol of...
-
Using data from Table 13.3, calculate the freezing and boiling points of each of the following solutions: (a) 0.25 m glucose in ethanol; (b) 20.0 g of decane, C10H22, in 50.0 g CHCl3; (c) 3.50 g NaOH...
-
Using data from Table 16.2, calculate the molar solubility of CaF2.
-
The National Health Statistics Reports dated Oct. 22, 2008, included the following information on the heights (in.) for non-Hispanic white females: a. Calculate and interpret a confidence interval at...
-
The income statement of Guesser Company is shown below. Additional information: 1. Accounts receivable decreased $63,000 during the year. 2. Inventory increased $38,000 during the year. 3. Prepaid...
-
Determine the size of the M1 money supply using the following information. Currency plus Travelers checks ... $25 million Negotiable CDs ........ $10 million Demand deposits ......... $13 million...
-
A parallel-plate capacitor with a plate area of \(50 \mathrm{~mm}^{2}\) and air between the plates can hold \(5.5 \mathrm{pC}\) of charge per volt of potential difference across its plates. When a...
-
G. Gabe, Attorney at Law, experienced the following transactions in 2009, the first year of operations. 1. Purchased $1,500 of office supplies on account. 2. Accepted $24,000 on February 1, 2009, as...
-
6. The number of (staircase) paths in the xy-plane from (0, 0) to (7, 5) where each such path is made up of individual steps going one unit upward (U) or one unit to the right (R). (a) 12C5 (b) 12!...
-
Prove that the moments of inertia I a and I b (see Figure 8.3) are equal for the benzene molecule. You do not need to know the bond distances, just invoke the hexagonal symmetry, and for simplicity,...
-
The barrier to rotation about the CC bond in CH 3 CH 2 Cl is about 15 to 20 kJ/mol. Estimate the torsional frequency and predict how it could be observed experimentally. Consult a table of bond...
-
What factors determine whether a person is an employee or an independent contractor?
-
Jeremy Hoven was employed as a pharmacist by Walgreen Company (Walgreens). Hoven experienced an armed robbery at a Walgreens store during his first year of work. Hoven asked Walgreens to increase...
-
3. Starting from this equation: - [U(X) U (XB,YB)] 8 < = Derive the probability of winning the election for candidate A that is given by the following equation: Pr (IIA > ) = Pr ( 8 < ) = + [, a'd'...
-
You will see that you will be asked to make three separate changes to the base scenario. These changes are noted on three separate spreadsheet worksheets (commonly called "tabs"). The first two...
-
Discuss the historical perspective, demand, and creation of the program. Include program benefits and who was covered. 2. Describe the historical and current lack of transparency in health care.
-
d) View the previous months' Accounts Receivable Action Plan below. Refer to Policies and Procedures and take note of the Actions Required. Action Required for Debtors Outstanding Greater Than 30...
-
Media experts claim that daily newspapers are declining because of Internet access. Listed below are the numbers of daily newspapers in the United States for a recent sequence of 20 years. Shown...
-
Some people argue that the internal control requirements of the Sarbanes-Oxley Act (SOX) put U.S. companies at a competitive disadvantage to companies outside the United States. Discuss the...
-
Identify the reagents that you would use to accomplish each of the following transformations: a. b. c. d. e. f. g. h. Br-
-
Consider the following two compounds. Monochlorination of one of these compounds produces twice as many stereoisomeric products as the other. Draw the products in each case, and identify which...
-
Draw a bond-line structure for each of the following compounds: a. CH 2 = CHCH 2 C(CH 3 ) 3 b. (CH 3 CH 2 ) 2 CHCH 2 CH 2 OH c. CH COCH 2 CH(CH 3 ) 2 d. CH 3 CH 2 OCH 2 CH 2 OCH 2 CH 3 e. (CH 3 CH 2...
-
What are the duties that must be fulfilled at all times when providing Financial Advice to a client?
-
Li is on an annual gross salary of $ 6 0 , 0 0 0 per annum. Resident Tax Rates 2 0 2 2 - 2 0 2 3 Taxable Income 1 0 - $ 1 8 , 2 0 0 $ 1 8 , 2 0 1 - $ 4 5 , 0 0 0 $ 4 5 , 0 0 1 - $ 1 2 0 , 0 0 0 $ 1 2...
-
A comparable property sold six months ago for $125,000. Market data indicates that the following adjustments are appropriate for the subject property Location adjustment +5% Time adjustment +6% Age...

Study smarter with the SolutionInn App


