Identify each of the properties illustrated in Problems 2732. a. a + (10 + b) = (a
Question:
Identify each of the properties illustrated in Problems 27–32.
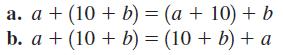
Transcribed Image Text:
a. a + (10 + b) = (a + 10) + b b. a + (10 + b) = (10 + b) + a
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a Ass...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify each of the properties illustrated in Problems 2732. x2+x-1 x2+x-1 x2-1 x2-1
-
Identify each of the properties illustrated in Problems 2732. 15 + [a + (-a)] = 15 + 0
-
Identify each of the properties illustrated in Problems 2732. a.mustard + catsup = catsup + mustard commutative b. (red + blue) + yellow = red + (blue + yellow)
-
A curve has equation y = x 2 ln 3x. Find the value of dy/dx and d 2 y/dx2 at the point where x = 2.
-
A glass ball of radius 2.00 cm sits at the bottom of a container of milk that has a density of 1.03 g/cm3. The normal force on the ball from the container's lower surface has magnitude 9.48 x 10-2 N....
-
A helium balloon lifts a basket and cargo of total weight 2000 N under standard conditions, in which the density of air is 1.29 kg/m3 and the density of helium is 0.178 kg/m3. What is the minimum...
-
P(170 < x < 195) The random variable x is normally distributed with mean = 174 and standard deviation = 20. Find the indicated probability.
-
Of the five profiled Intel CEOs, which would you characterize as the most job centered leader? The least job centered? The most employee centered leader? The least employee centered? Whose style most...
-
Discuss the challenges and opportunities of scaling up process intensification concepts from laboratory-scale experiments to industrial-scale applications, addressing issues related to...
-
Find Figure 5.18 shows the cover of a leading mathematics journal. It depicts the symbols for the twelve animals in the Chinese zodiac. (2013, for example, is the year of the snake and 2014 is the...
-
Identify each of the properties illustrated in Problems 2732. a. 5 1 = 1 5 b. 3(4 + 8) = 3 (4) + 3(8)
-
Partial regression results from a sample of 13 observations are shown below. Fill in the three missing values indicated by ()*. Is the coefficient for the variable x (9.16) statistically significant...
-
You draw in a deep breath on a chilly day, inhaling 3.0 L of 0C air. If the pressure in your lungs is a constant 1.0 atm, how much heat must your body supply to warm the air to your 37C internal body...
-
Suppose you start your day in Denver, on a cool 10C spring day. The local atmospheric pressure is 85 kPa. You fill your cars tires until the gauge shows 210 kPa (about 30 psi). You then drive up to...
-
In the chapter, you saw that bridges often have expansion joints to account for the changes in size due to temperature changes. Suppose a bridge is supported by steel girders; the central span has a...
-
Its possible to boil water by adding hot rocks to it, a technique that has been used in many societies over time. If you heat a rock in the fire, you can easily get it to a temperature of 500C. If...
-
Your heart does work to pump blood through your body. Each minute, 5.0 L of blood travels through your circulatory system. The pressure drops from 16 kPa as it exits the heart to approximately zero...
-
What does it mean when a variance is favorable? What does it mean when a variance is unfavorable?
-
After looking at the resources, explain what a spirit image is. Why might looking at a god and/or a human in terms of their spirit be helpful if you want to eliminate some of the divisions between...
-
Write a balanced equation for each of these cases: a) The reaction between sodium metal and bromine gas. b) The reaction between potassium metal and water. c) The reaction between hydrogen gas and...
-
Determine whether the bond between each pair of atoms will be pure covalent, polar covalent or ionic: a) S and O b) Al and F c) C and Br d) Mg and Cl
-
Write a Lewis structure for CH 3 Cl.
-
xPlease address the questions below in a memorandum to Apex Investment Partners. Please include your revised capitalization tables and waterfall diagrams. How, if at all, would you change the...
-
Sky Company employed Tom Mills in Year 1. Tom earned $5,700 per month and worked the entire year. Assume the Social Security tax rate is 6 percent for the first $110,000 of earnings, and the Medicare...
-
Pioli Corporation manufactures one product. It does not maintain any beginning or ending Work in Process inventories. The company uses a standard cost system in which inventories are recorded at...

Study smarter with the SolutionInn App


