Identify each of the properties illustrated in Problems 2732. x2+x-1 x2+x-1 x2-1 x2-1
Question:
Identify each of the properties illustrated in Problems 27–32.
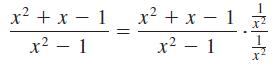
Transcribed Image Text:
x2+x-1 x2+x-1 x2-1 x2-1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

Aqib Parvej
I am teaching since my graduation time so I have teaching experience of about 5 years and in these years I learn to teach in the best and interesting way .
4.80+
20+ Reviews
41+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify each of the properties illustrated in Problems 2732. 15 + [a + (-a)] = 15 + 0
-
Identify each of the properties illustrated in Problems 2732. a.mustard + catsup = catsup + mustard commutative b. (red + blue) + yellow = red + (blue + yellow)
-
Identify each of the properties illustrated in Problems 2732. a. a + (10 + b) = (a + 10) + b b. a + (10 + b) = (10 + b) + a
-
Laura Leasing SA signs an agreement on January 1, 2022, to lease equipment to Plote AG. The following information relates to this agreement. 1. The term of the non-cancelable lease is 3 years with no...
-
Caught in an avalanche, a skier is fully submerged in flowing snow of density 96 kg/m3. Assume that the average density of the skier, clothing, and skiing equipment is 1020 kg/m3. What percentage of...
-
Design a roller chain to transmit power from a 20 kW motor to a reciprocating pump. The pump is to operate continuously 24 hours per day. The speed of the motor is 600 r.p.m. and that of the pump is...
-
The ages of prime ministers of Sri Lanka, when they were first sworn to office, are normally distributed with a mean of 58.21 years and a standard deviation of 11.56 years. Find the probability that...
-
Thomas Corporation makes bicycles. It has always purchased its bicycle tires from the Firelock Company at $12 each, but is currently considering making the tires in its own factory. The estimated...
-
A soft tennis ball is dropped onto a hard floor from a height of 1.55 m and rebounds to a height of 1.12m. (Assume that the positive direction is upward.) (a) Calculate its velocity (in m/s) just...
-
Find Figure 5.18 shows the cover of a leading mathematics journal. It depicts the symbols for the twelve animals in the Chinese zodiac. (2013, for example, is the year of the snake and 2014 is the...
-
Identify each of the properties illustrated in Problems 2732. a. 5 1 = 1 5 b. 3(4 + 8) = 3 (4) + 3(8)
-
Solid carbon can react with gaseous water to form carbon monoxide gas and hydrogen gas. The equilibrium constant for the reaction at 700.0 K is K p = 1.60 * 10 -3 . If a 1.55-L reaction vessel...
-
Humans can produce an output power as great as 20 W/kg during extreme exercise. Sloths are not so energetic. At its maximum speed, a 4.0 kg sloth can climb a height of 6.0 m in 2.0 min. Whats the...
-
The opposite of a wind turbine is an electric fan: The electric energy that powers the fan is converted to the kinetic energy of moving air. A fan is putting 1.0 J of kinetic energy into the air...
-
A typical meteor that hits the earths upper atmosphere has a mass of only 2.5 g, about the same as a penny, but it is moving at an impressive 40 km/s. As the meteor slows, the resulting thermal...
-
Most of the energy you expend in cycling is dissipated by the drag force. If you double your speed, you increase the drag force by a factor of 4. This increases the power to cycle at this greater...
-
You can, in an emergency, start a manual transmission car by putting it in neutral, letting the car roll down a hill to pick up speed, then putting it in gear and quickly letting out the clutch. If...
-
What are the three primary reasons variances occur?
-
If a test has high reliability. O the test measures what the authors of the test claim it measures O people who take the same test twice get approximately the same scores both times O scores on the...
-
A cigarette lighter contains 2.90 g of butane, C 4 H 10 . Calculate the heat (in kJ) associated with the complete combustion of the butane in the lighter. C 4 H 10 (g) + 13/2 O 2 (g) 4 CO 2 (g) + 5...
-
Calcium metal reacts with hydrochloric acid according to the balanced equation: Ca(s) + 2 HCl (aq) CaCl 2 (aq) + H 2 (g) A 0.150 g sample of Ca metal is combined with enough HCl to make 100.0 mL of...
-
Use the standard enthalpies of formation to determine H rxn for the reaction: Zn(s) + 2 HCl(aq) ZnCl 2 (aq) + H 2 (g)
-
McCullough Hospital uses a job-order costing system to assign costs to its patients. Its direct materials include a variety of items suc as pharmaceutical drugs, heart valves, artificial hips, and...
-
Queens and Kings Inc. sells high quality chess sets (with a clock) for $100.00. The fixed costs for the business are $120,000 and the variable cost per set is $60.00. Determine: (7 marks total) a....
-
The accounting records of Wall's China Shop reflected the following balances as of January 1, Year 2. Cash Beginning inventory $ 18,900 13,795 (155 units @ $89) 15,900 Common stock Retained earnings...

Study smarter with the SolutionInn App


