Use this information to answer the questions in Problems 18-23. a. What does the notation (DACB) mean?
Question:
Use this information to answer the questions in Problems 18-23.
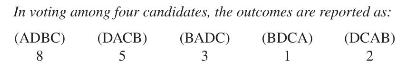
a. What does the notation (DACB) mean?
b. What does the " 5 " under (DACB) mean?
Transcribed Image Text:
In voting among four candidates, the outcomes are reported as: (DACB) 5 (ADBC) 8 (BADC) 3 (BDCA) 1 (DCAB) 2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-6. On December 12, Irene purchased the building where her store is located. She paid...
-
Use this information to answer the questions in Problems 12-17. a. What does the notation (CAB) mean? b. What does the " 5 " under (CBA) mean? In voting among three candidates, the outcomes are...
-
Use this information to answer the questions in Problems 18-23. a. What does the notation (ADBC) mean? b. What does the " 8 " under (ADBC) mean? In voting among four candidates, the outcomes are...
-
public class Class extends ClassA public classB ( ( Time left 0 0 : 1 2 : 0 3 int init = 1 0 ; super ( 4 0 ) ; O a . . The method super is not defined. b . . No values may be passed to super. O c . ....
-
A popular brand of cola contains 6.50 g of carbon dioxide dissolved in 1.00 L of soft drink. If the evaporating carbon dioxide is trapped in a cylinder at 1.00 atm and 20.0C, what volume does the gas...
-
Sticky Fingers Inc. produces scotch tape and masking tape. Last year's annual report has been compiled, and you are in charge of business analysis for the year. The company had a goal for inventory...
-
Blade efficiency is the ratio of: (a) Work done of blades and energy supplied to the blades (b) Work done on blade and energy supplied to each stage (c) Energy supplied per stage and work done on the...
-
Choose the best answer. 1. Under the hierarchy of GAAP for a state and local government, which of the following has the highest level of authority? a. AICPA Practice bulletins if specifically made...
-
Name four characteristics of Force?
-
Find the standard divisor (to two decimal places) for the given populations and number of representative seats in Problems 15-22. Population 8,920,000 # Seats 12
-
The Adobe School District is hiring a vice principal and has interviewed four candidates: Andrew (A), Bono (B), Carol (C), and Davy (D). The hiring committee members have indicated their preferences:...
-
At a certain Noodles & Company restaurant, customers arrive during the lunch hour at a rate of 2.8 per minute. What is the probability that (a) At least 30 seconds will pass before the next customer...
-
Ivy begins trading as a farmer on 1 January 2019, making up annual accounts to 31 December. Her adjusted trading profits/(losses) in the opening years are as follows: (a) Compute Ivy's trading income...
-
For each of the following taxpayers, state the dates on which the 2021-22 income tax is due to be paid and calculate the amount payable on each date. (Ignore Class 4 NICs). (a) Guy's 2020-21 income...
-
Julian, a self-employed shopkeeper, takes goods costing 30 from his trading stock for his own personal use. If he had sold the stock to a customer he would have charged 45. When computing his trading...
-
(a) As from 1 January 2022, a motor car with a retail price of 21,000 is leased for four years at a cost of 300 per month. The car is used only for trade purposes and has an emission rating of...
-
Jabran did not receive a notice to file a tax return for 2020-21 but he was aware that he had income which had not been assessed to tax. He notified HMRC of this fact on 2 October 2021 and a return...
-
You are considering opening a bagel restaurant aimed primarily at the breakfast trade. Youll sell bagels, coffee, and other items in relatively fixed proportions to one another. For each bagel sold,...
-
A container holds 2.0 mol of gas. The total average kinetic energy of the gas molecules in the container is equal to the kinetic energy of an 8.0 10-3-kg bullet with a speed of 770 m/s. What is the...
-
Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here. H,N* NH2 NH2 NH C=0 CH2 CH2 S CH2 CH2 CH2 CH2 (a) HCHCO, (b) H3CHCO, (c) H,NCHCO, Glutamine Cysteine Arginine
-
Phosphate at 0.01 M is one of the main buffers in blood plasma, whose pH is 7.45. Would phosphate be as useful if the plasma pH were 8.5?
-
Find the pH and the concentration of each species of lysine in a solution of 0.010 0 M lysine ? HCl, lysine monohydrochloride. The notation lysine ? HCl refers to a neutral lysine molecule that has...
-
Identify workplace tasks that involve hazardous chemicals e.g.: manufacturing cleaning painting Use guidance materials to determine the potential worker exposure to the identified hazardous chemicals...
-
Describe how organizations can effectively navigate resistance to change among employees and foster a culture that embraces and adapts to change. Provide specific examples from real-world cases or...
-
There are four regions of a certain country that meet at a common corner. The areas of the four regions, in square miles, are shown in the table. Region 1 71,723 (a) Mentally estimate the combined...

Study smarter with the SolutionInn App


