Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here.
Question:
Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here.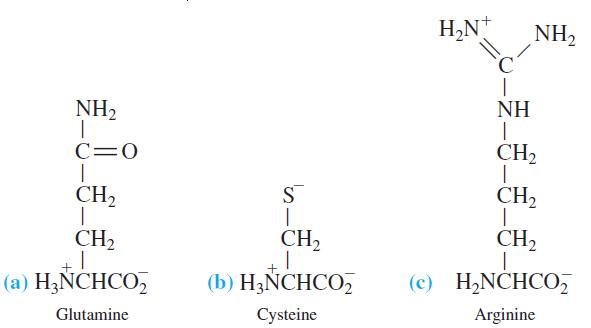
Transcribed Image Text:
H,N* NH2 NH2 NH C=0 CH2 CH2 S CH2 CH2 CH2 CH2 (a) H¿ÑCHCO, (b) H3ÑCHCO, (c) H,NCHCO, Glutamine Cysteine Arginine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Amina acid PH PH of each Solution 0010 M The three forms of glutamine is as follows L...View the full answer

Answered By

Manoj Kumar Mahariya
Teaching experience-
1.As a lecture in Akash Carrer institute (since 2 year)
2.As a assistant lecturer in National Institute of technology,Punjab
3.Also school lecture of pcbm subject in Ravindra manorial senior sec.school, Rajasthan
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the pH of a solution prepared by (a) Dissolving 36.5 g of lactic acid in water and diluting to 500 mL. (b) Diluting 25.0 mL of the solution in (a) to 250 mL. (c) Diluting 10.0 mL of the...
-
Calculate the pH of a solution made by mixing 0.60 L of 0.10 M NH4Cl with 0.50 L of 0.10 M NaOH. Kb for NH3 is 1.8 10-5.
-
Calculate the pH of a solution made by mixing 7.52 mL of 4.9 10-2 M Ca(OH)2 with 22.5 mL of 0.11 M HCl.
-
The following information was taken from the annual manufacturing overhead cost budget of Granada Company. Variable manufacturing overhead costs .....$33,000 Fixed manufacturing overhead costs...
-
The GSS asked respondents to report their opinion on spanking as a method to discipline a child (SPANKING). Examine how respondents' attitudes toward spanking a child are associated with SEX, CLASS,...
-
Stock in Bunyan Brewery is currently priced at $20 per share. A call option with a $20 strike price and 60 days to maturity is quoted at $2. Compare the percentage gains and losses from a $2,000...
-
What are three major cost flow assumptions used by U.S. companies in valuing inventory? (a) LIFO, FIFO, average market. (b) LIFO, FIFO, actual cost. (c) LIFO, FIFO, average cost. (d) LIFO, FIFO,...
-
Matthew Liotines Dream Store sells beds and assorted supplies. His best- selling bed has an annual demand of 400 units. Ordering cost is $ 40; holding cost is $ 5 per unit per year. a) To minimize...
-
Natural Colour Co. wants to invest in a bond with par value of $ 1000 that offers 10% coupons paid semi-annually. The bond has 15 years to maturity and has a yield to maturity of 9%. If the current...
-
LibraryPreston Library, a nonprofit organization, presented the following statement of financial position and statementof activities for its fiscal year ended February 28, 2014.Preston...
-
Interpreting spectral data. The graph shows the 1H-nuclear magnetic resonance chemical shift of the H 4 proton on pyridine as a function of pH. Chemical shift is related to the environment of a...
-
Phosphate at 0.01 M is one of the main buffers in blood plasma, whose pH is 7.45. Would phosphate be as useful if the plasma pH were 8.5?
-
Propose an efficient synthesis for each of the following transformations: (a) (b) (c) (d) (e) (f) (g) -OH
-
This week as Menifield described the importance of effectively communicating data, we learned the critical importance of how data is represented in presentations. As you learned, data can be...
-
solve the following lim I-9 7+37 27 15 is
-
Why are measure of location, commonly called measures of center, not sufficient in themselves to get a good picture of a data set? Support your answers with an example of a business application.
-
According to American School Search.com, HutchCC's current retention rate (that is, students who return to HutchCC) is 55%. The graduation rate is 33%. In your opinion, how could HutchCC improve...
-
Friday's snowfall was 18 cm and Saturday's snowfall was 21 cm. What must be Sunday's snowfall to set a weekly average record of 16 cm?
-
A grating has 5000.0 slits / cm. How many orders of violet light of wavelength 412 nm can be observed with this grating?
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
A 10.0-mL solution of 0.050 0 M AgNO 3 was titrated with 0.025 0 M NaBr in the cell S.C.E. 7 || titration solution | Ag(s) Find the cell voltage for 0.1 and 30.0 mL of titrant.
-
A solution containing 50.0 mL of 0.100 M EDTA buffered to pH 10.00 was titrated with 50.0 mL of 0.020 0 M Hg(ClO 4 ) 2 in the cell shown in Exercise 14-B: S.C.E. 7 titration solution | Hg(l) From the...
-
Consider the titration in Figure 15-2. (a) Write a balanced titration reaction. (b) Write two different half-reactions for the indicator electrode. (c) Write two different Nernst equations for the...
-
Current home mortgage interest rates are approximately 4% for a 10 year loan and 3% for a 8 year loan. Which annual payment (A) has a lower present worth (P)?
-
Comparison rates in credit advertisements for fixed term credit for personal domestic household purposes must be made available. Select one: True False
-
Results from a regression model analysis are shown below. The numbers below the coefficient estimates are the sample standard errors of the coefficient estimates. a. Compute two-sided 95% confidence...

Study smarter with the SolutionInn App


