Deduce the structures of compounds EL in the roadmap below. E (CH13 Br) HBr (no peroxides) F
Question:
Deduce the structures of compounds E–L in the roadmap below.
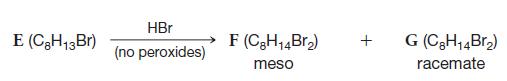
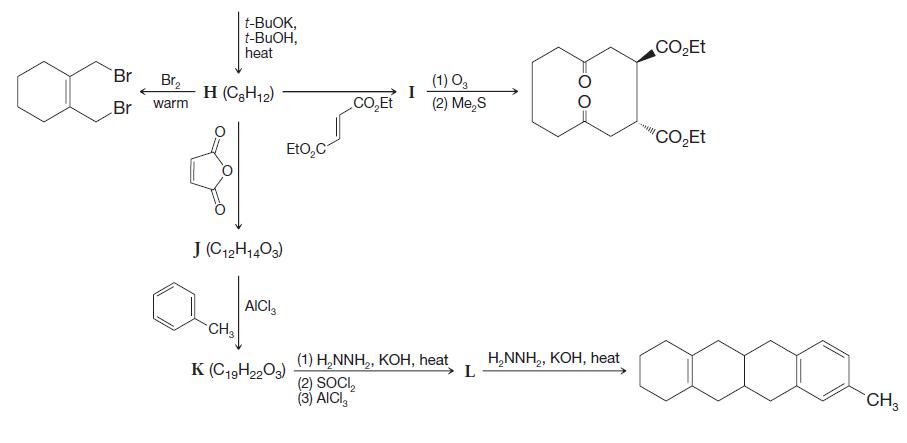
Transcribed Image Text:
E (C₂H13 Br) HBr (no peroxides) F (C8H₁4Br₂) meso + G (C₂H₁4Br₂) racemate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Answer Compound E C6H14 Compo...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Using the information provided below, deduce the structures of compounds A, B, C, and D: 1) EtMgBr 1) O3 (C10H12) (C,H160) 2) H,0 2) DMS (C9H100) [H*), (CH3)2NH AICI3 (-H20)
-
Deduce the structures of the following compounds. (a) C4H8O: IR 1717 cm-1 NMR 0.95 (3H, t, J = 8 Hz); 2.03 (3H, s): 2.38 (2H. q, J = 8 Hz) (b) A compound with molecular mass = 70.1, IR absorption...
-
Deduce the structures of compounds AD. Draw structures that show stereochemistry where appropriate: CgH1,04 D OH (optically inactive) (1) hot KMNO,, HO | (2) H,0+ CH18 (1) Li, EINH, (2) NH,CI H2,...
-
There is a crop that the value p next month is random. The value can be either small p=1 or large P=3 depending on the weather. Half of people are optimists believing the value of the crop will be...
-
6p + 2 /8 4p 1 /6 Describe the solution set as an inequality, in interval notation, and on a graph.
-
An experiment involves tossing a pair of dice, 1 green and 1 red, and recording the numbers that come up. If x equals the outcome on the green die and y the outcome on the red die, describe the...
-
Label each of the following characteristics of a corporation as either an (A) advantage or a (D) disadvantage: a. Organizational costs b. Continuity of existence c. Capital raising capability d....
-
Texas Roadhouse opened a new restaurant in October. During its first three months of operation, the restaurant sold gift cards in various amounts totaling $3,500. The cards are redeemable for meals...
-
Problem 8-33 Equivalent Annual Annuity (LO5) A firm can lease a truck for 4 years at a cost of $36,000 annually. It can instead buy a truck at a cost of $86,000, with annual maintenance expenses of...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
Synthesize 2-chloro-4-nitrobenzoic acid from toluene and any other reagents necessary. Begin by writing a retrosynthetic analysis.
-
Elucidate the structures of compounds A through I in the following road map problem. Specify any missing reagents. A (C5H) Br., warm (1 molar equiv.) F CH,ONA (2 molar equiv.) G 1 HBr (no ROOR) H...
-
How might we distinguish 3PLs from other LSPs?
-
A corporation is listed on the New York Stock Exchange. Unfortunately, it has seen hard times, and its stock price has dropped to $0.75 per share. Unless its stock trades at above $1.00, it may be...
-
Bill Stale started investing in the stock market this year. He bought two stocks and sold them both just before the end of the financial year. Stock A made was sold at a $400 gain and Stock B sold at...
-
A start-up company plans to pay a $0.20 dividend in one year and grow that dividend at 10% per year for the following five years (up through the sixth dividend). Starting in the seventh year, the...
-
Identifying how a company performs a market analysis. 2. Confirm the company's e-Marketing goals. 3. How they evaluate electronic communications. 4. Identifying how a company identifies and reaches...
-
Your borrowers have monthly income of $15,000, in the neighborhood that they are looking to purchase the average property tax per year is $4,800 and the average heat cost per year is $2,400. Their...
-
A portfolio is invested 20 percent in Stock G, 35 percent in Stock J, and 45 percent in Stock K. The expected returns on these stocks are 9.2 percent, 12 percent, and 15.7 percent, respectively. What...
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
Draw both chair conformations of 1-methyl-1-phenylcyclohexane. Which is more stable by how much energy?
-
Explain whether the methyl is axial or equatorial in this compound: CH3 H C-C-CH3 CH3
-
Draw both chair conformations for menthol (a component of peppermint oil) and its stereo isomer, neo menthol. Which groups are axial and which groups are equatorial? Explain which conformation is...
-
Accumulations of shifting sand are Question 42 options: spurs. dunes. horns. cirques
-
Beating the competition; European Wax Centers (EWC) and Massage Envy---identifies option for the EWC to beat their competition based upon concepts learned in BUSU 630. Provides a clear picture of the...
-
P5.6B During the past year, Stacy Clark planted a new orange grove on 250 acres of land that she leases for $45,000 a year. She has asked you, as her accountant, to assist her in determining the...

Study smarter with the SolutionInn App


