Papaverine has been synthesized by the following route: Outline the reactions involved. CHO CHO NH CHO CHO
Question:
Papaverine has been synthesized by the following route:
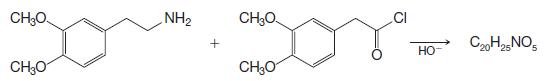

Outline the reactions involved.
Transcribed Image Text:
CH₂O CH₂O NH₂ CH₂O CH₂O CI НО- C20H25 NO5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Solution The reaction that takes place is as follows In the first step hydroxyl OH ions are form...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
The sex hormone estrone has been synthesized by a route that involves the following step. Identify the pericyclic reactions involved, and propose amechanism. CH CH Heat |CH30 CH0 Estrone methyl ether
-
Gramin has been synthesized by heating a mixture of indole, formaldehyde, and dimethylamine. (a) What general reaction is involved here? (b) Outline a reasonable mechanism for the gramine synthesis.
-
The sedative-hypnotic ethinamate can be synthesized by the following route. Provide structures for ethinamate and the intermediates A and B: Cyclohexanone (1) HC=CNa, (2) HO+ A (CH12O) CICOCI B...
-
Due to improved technology and public-service campaigns, the number of collisions at highwayrailroad crossings per year has declined since 1992 (see Table 52). Let n be the number of collisions (in...
-
Ford Motor Co. is considering alternate methods of accounting for the cash discounts it takes when paying suppliers promptly. One method suggested was to report these discounts as financial income...
-
A distillation column at \(101.3 \mathrm{kPa}\) is separating a two-phase feed that is \(60.0 \%\) liquid, \(40.0 \mathrm{~mol} \%\) methanol, and \(60.0 \mathrm{~mol} \%\) water. Distillate product...
-
Consider the following information from Alliance Data Systems Corporation 2009 10K. On October 30, 2009, the Company assumed the operations of the Charming Shoppes' credit card program, including the...
-
Systems Understanding Aid - COSO Framework Requirement: Evaluate the Waren Company using the COSO framework as a tool. The Waren Company flowcharts will be incredibly helpful for this exercise. The...
-
A quarterback on a football team has a pass completion rate of 0.62. If, in a given game, he attempts 16 passes, what is the probability that he will complete (a) 12 passes? (b) More than half of his...
-
One of the important steps in Gates synthesis of morphine involved the following transformation: Suggest how this step was accomplished. CHO CHO NC CHO CHO NC-
-
Many alkaloids appear to be synthesized in plants by reactions that resemble the Mannich reaction. Recognition of this (by R. Robinson in 1917) led to a synthesis of tropinone that takes place under...
-
The government has undertaken a highway project that was originally projected to cost $1 billion and provide benefits of $1.5 billion. Unfortunately, the costs have been much higher than anticipated....
-
Jean Munyutu is an African student who went to France on a trip to study to be a pastry chef. He met and became friends with Francois Dubois. After spending some time together and becoming friends,...
-
XYZ Corporation's next dividend is expected to be $3 per share. Dividend growth rate has been at 2% and expected to be so into the future. If investor's return is 10%, calculate the stock price in...
-
Automobili Lamborghini S.P.A. v Lamborghini Latino America USA 400 F.Supp.3d 471 (E.D. Va. 2019) Facts Jorge Antonio FernandezGarcia, who lived in South America, told Anthony Crudup (defendants), who...
-
Under what circumstances would absorption costing and variable costing produce different results in terms of net income, and what are the implications for decision-making and financial reporting? You...
-
The concept of Cost Control and Budgeting: Which of the budgets do you think may be the most important in a company? Do you think many of those would translate well into your personal finances? Are...
-
A retail company recently completed a physical count of ending merchandise inventory to use in preparing adjusting entries. In determining the cost of the counted inventory, company employees failed...
-
A condenser (heat exchanger) brings 1 kg/s water flow at 10 kPa quality 95% to saturated liquid at 10 kPa, as shown in Fig. P4.91. The cooling is done by lake water at 20C that returns to the lake at...
-
Determine the structure of this compound from its IR and 13C-NMR spectra, its formula isC7H16O2: C,H1,0, 80- 60- %T 40 20- 0- 500 1000 1500 2000 2500 3500 3000 4000 Wavenumber (cm) C,H1602 All CH's...
-
Predict the multiplicities of the indicated hydrogen's in the 1H-NMR spectra of these compounds: CH,CI I H I - + CH,CI CI - c) b) --- I H I - ()
-
Predict the approximate chemical shifts, multiplicities and integrals for the absorptions in the 1H-NMR spectra of thesecompounds: CH2 b) .- COCH CH3 a) d) CH;CHCH CH3 c) CH;CH,NHCH,CH3
-
Technology Fund X weight (%) 32.2 2. The mutual fund X has value weights on four sectors and returns as given in the table below. The table also shows the weights and returns of the fund's benchmark...
-
eBook Problem 7 - 0 6 A widow currently has a $ 8 2 , 0 0 0 investment that yields 7 percent annually. Can she withdraw $ 2 1 , 0 0 0 for the next five years? Use Appendix D to answer the question....
-
Charleston Affair uses the product cost concept to price its goods. The company plans to release a new product in the upcoming month. Use the information shown below to determine: (A) Product cost...

Study smarter with the SolutionInn App


